| Synonym: | Sulfo-N-succinimidyl 4-maleimidobutyrate sodium salt |
| CAS #: | 185332-92-7 |
| Molecular Formula: | C12H11N2NaO9S |
| Molecular Weight: | 382.3 |
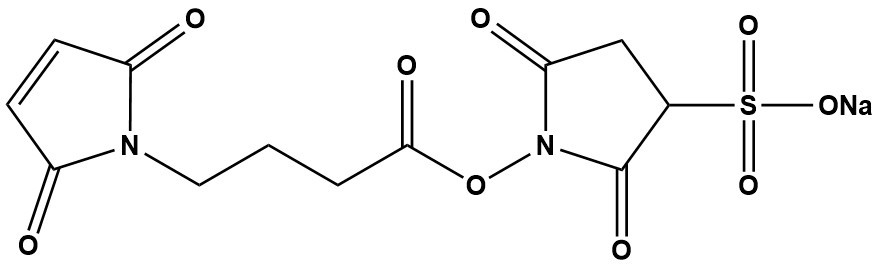
| Sulfo-GMBS is a water-soluble, heterobifunctional crosslinking reagent widely used in biochemistry, immunology, and bioconjugation. It belongs to a critical class of tools that enable the covalent coupling of two different molecules or entities, typically a protein (like an antibody) and another molecule (like a toxin, enzyme, or solid support). The “heterobifunctional” nature is its key feature, meaning it possesses two different reactive groups at opposite ends of its molecular spacer arm. This allows for a controlled, stepwise conjugation process, minimizing homodimerization (e.g., antibody-antibody coupling) which is a common problem with homobifunctional crosslinkers. 1. Chemical Structure and Reactive Groups The structure of Sulfo-GMBS can be broken down into three parts: • NHS Ester (Sulfo-NHS Ester): This is the amine-reactive end. Specifically, it reacts efficiently with primary amines (ε-amino group of lysine residues or the N-terminus of proteins) to form stable amide bonds. The “Sulfo” prefix indicates a sulfonate group (-SO₃⁻) on the N-hydroxysuccinimide ring. This charged group dramatically increases the water solubility of the reagent compared to its non-sulfonated counterpart, GMBS. • Maleimide Group: This is the thiol-reactive end. It specifically and rapidly reacts with free sulfhydryl groups (-SH) from cysteine residues to form stable thioether bonds. Maleimide-thiol reactions are highly selective under neutral to slightly alkaline conditions (pH 6.5-7.5). • Spacer Arm: The two reactive groups are separated by a butyryl spacer arm, which has a defined chain length. This spacer (approximately 10.4 Å) provides sufficient distance to avoid steric hindrance between the two molecules being conjugated. Chemical Reaction Summary: • Step 1 (Amine Coupling): Sulfo-GMBS is first incubated with a molecule containing primary amines (e.g., an antibody). The Sulfo-NHS ester reacts to form an amide bond, creating a “maleimide-activated” protein. • Step 2 (Thiol Coupling): This activated protein is then purified to remove excess, unreacted crosslinker. Finally, it is mixed with a thiol-containing molecule (e.g., a cysteine-containing peptide, a reduced antibody, or a protein with an engineered cysteine). The maleimide group reacts to form the final, stable conjugate. 2. Key Properties and Characteristics • Solubility: Highly soluble in water and aqueous buffers (PBS, HEPES, etc.). This is its primary advantage over GMBS, which requires dissolution in organic solvents like DMSO or DMF that can be denaturing for some proteins. • Specificity: High specificity for amines (via NHS-ester) and thiols (via maleimide). This allows for precise targeting of common functional groups in biomolecules. • Stability: The dry powder should be stored desiccated at ≤ -20°C, protected from light and moisture. In solution, the Sulfo-NHS ester is highly susceptible to hydrolysis, especially at higher pH. The maleimide group can also hydrolyze to a non-reactive maleamic acid over time. Therefore, solutions must be prepared fresh and used immediately. • Membrane Impermeability: Due to the charged sulfonate group, Sulfo-GMBS is non-membrane-permeable. This makes it unsuitable for labeling intracellular targets in live cells but ideal for cell surface labeling or crosslinking extracellular proteins without affecting internal processes. 3. Advantages and Strengths • Controlled, Two-Step Conjugation: The primary advantage is the ability to perform a sequential, controlled reaction, drastically reducing unwanted homopolymer formation. • Excellent Water Solubility: Eliminates the need for organic solvents, simplifying protocols and preserving protein integrity. • High Efficiency and Specificity: The maleimide-thiol reaction is one of the most efficient and specific in bioconjugation chemistry. • Well-Defined Spacer: The spacer arm is long enough to be effective but short enough to avoid unnecessary flexibility that could lead to non-specific interactions. • Proven Track Record: A classic, well-characterized reagent with extensive literature support for protocols involving antibody-drug conjugates (ADCs), enzyme immobilization, and hapten-carrier protein conjugation. 4. Limitations and Considerations • Hydrolysis Sensitivity: Both reactive groups are labile in aqueous solution. The NHS ester has a half-life of minutes to hours depending on pH and temperature. This necessitates careful timing and buffer preparation. • Competition with Disulfide Bonds: If the target protein has existing disulfide bonds, they may need to be reduced (e.g., with DTT or TCEP) to generate free thiols for the maleimide to react with. This reduction step must be carefully controlled and the reducing agent must be thoroughly removed before the conjugation step. • Potential for Maleimide Hydrolysis/Exchange: While the thioether bond is stable, the maleimide ring can undergo hydrolysis after conjugation. In vivo, there is a potential for thiol exchange with glutathione, which can lead to deconjugation, a consideration for therapeutic ADCs. (This has led to the development of next-generation maleimides). • Amine Availability: The efficiency of the first step depends on the availability and accessibility of lysine residues on the target protein. 5. Comparison to Related Reagents • vs. GMBS: Sulfo-GMBS is the water-soluble version of GMBS. GMBS requires organic solvents, making it less convenient for direct use with sensitive proteins. • vs. SMCC / Sulfo-SMCC: SMCC is a very similar heterobifunctional crosslinker with the same reactive groups. The key difference is the spacer arm; SMCC has a longer and more rigid cyclohexane ring in its spacer, making it slightly longer and potentially more stable. The choice between GMBS and SMCC is often empirical. • vs. Homobifunctional Linkers (e.g., BS³, DTSSP): Homobifunctional NHS-esters cause uncontrolled polymerization, whereas Sulfo-GMBS provides directed, specific conjugation. • vs. Click Chemistry Reagents: Modern click chemistry (e.g., azide-alkyne cycloaddition) offers superior orthogonality and bioorthogonality but often requires genetic engineering of the target proteins to incorporate non-natural functional groups. 6. Typical Applications Sulfo-GMBS is a workhorse reagent in numerous applications: • Antibody-Drug Conjugate (ADC) Synthesis: Conjugating cytotoxic drugs (often thiol-modified) to monoclonal antibodies. • Enzyme-Protein/Peptide Conjugates: Creating immunogens for antibody production by coupling a hapten (small peptide) to a large carrier protein like KLH or BSA. • Immobilization of Biomolecules: Covalently attaching proteins, antibodies, or enzymes to thiol-activated surfaces, beads, or biosensors. • Cell Surface Labeling: Labeling surface-exposed proteins on cells for tracking or isolation, without penetrating the cell membrane. • Structure-Function Studies: Crosslinking interacting proteins to study complex formation and protein-protein interactions. 7. Storage and Handling • Storage: Store desiccated at -20°C or below. Protect from light and moisture. Allow the vial to warm to room temperature before opening to prevent condensation. • Solution Preparation: Prepare a fresh solution in a suitable amine-free buffer (e.g., PBS, pH 7.2-7.5) immediately before use. Do not store stock solutions. 8. Typical Conjugation Protocol • Step 1: Incubate your amine-containing protein (e.g., Antibody) with a ~10-20 molar excess of Sulfo-GMBS for 30-60 minutes at room temperature. • Purification: Use a desalting column (e.g., PD-10) or dialysis to rapidly remove excess, unreacted crosslinker and hydrolysis byproducts. This step is critical. • Step 2: Immediately mix the maleimide-activated protein with your thiol-containing molecule. Incubate for 1-2 hours at room temperature or overnight at 4°C. • Quenching & Final Purification: A small excess of a free thiol (e.g., 2-mercaptoethanol, cysteine) can be added to quench any unreacted maleimide groups. Purify the final conjugate using size-exclusion chromatography or dialysis. Sulfo-GMBS (CAS 185332-92-7) remains a fundamental and highly valuable tool in the bioconjugation toolkit. Its heterobifunctional design, combined with excellent water solubility, provides a robust method for creating defined conjugates between amines and thiols. While its sensitivity to hydrolysis requires careful handling, its proven efficacy, specificity, and the controlled nature of its reactions make it a top choice for applications ranging from immunoassay development and ADC research to general protein immobilization. For any scientist requiring a reliable, water-soluble crosslinker for amine-to-thiol coupling, Sulfo-GMBS is an excellent and time-tested option. |
|
Sulfo-GMBS
For Research & Development use only. Not for testing and/or use on humans.