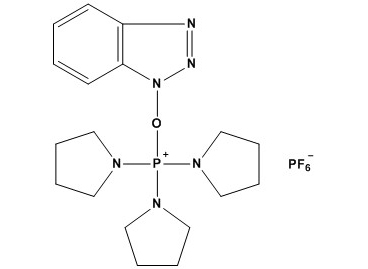
| Synonym: | Benzotriazole-1-yl-oxy-tris-pyrrolidino-phosphonium hexafluorophosphate |
| CAS #: | 128625-52-5 |
| Molecular Formula: | C18H28F6N6OP2 |
| Molecular Weight: | 520.4 |
| 1. Introduction and Chemical Identity PyBOP (CAS #: 128625-52-5) is a highly efficient peptide coupling reagent that has become an indispensable tool in modern organic synthesis, particularly in peptide chemistry and the formation of amide bonds. Since its introduction in the 1990s, PyBOP has gained widespread acceptance in both academic and industrial settings due to its superior reactivity, minimal racemization, and excellent compatibility with a wide range of functional groups. IUPAC Name: Benzotriazol-1-yl-oxytripyrrolidinophosphonium hexafluorophosphate Molecular Formula: C18H28F6N6OP2 Molecular Weight: 520.4 g/mol CAS Number: 128625-52-5 Appearance: White to off-white crystalline powder Melting Point: 145-150°C (decomposition) Solubility: Highly soluble in DMF, DCM, acetonitrile; insoluble in water Storage Conditions: Store at 2-8°C under inert atmosphere, protect from moisture 2. Mechanism of Action PyBOP functions as an efficient coupling reagent through a well-established mechanism that involves the formation of a highly reactive phosphonium-activated intermediate. Understanding this mechanism is crucial for optimizing reaction conditions and troubleshooting coupling failures. 2.1 General Coupling Mechanism The coupling reaction proceeds through the following key steps: Step 1: Activation of the Carboxylic Acid In the presence of a non-nucleophilic base (typically DIPEA or N-methylmorpholine), the carboxylic acid is deprotonated to form a carboxylate anion. This anion then attacks the electron-deficient phosphorus center of PyBOP, displacing the benzotriazoloxy (BtO) leaving group. This results in the formation of an acyloxyphosphonium intermediate, which is the key activated species. Step 2: Nucleophilic Attack by the Amine The amine nucleophile attacks the carbonyl carbon of the acyloxyphosphonium intermediate. This step is facilitated by the excellent leaving group ability of the tripyrrolidinophosphine oxide moiety. The tetrahedral intermediate collapses to expel the phosphine oxide, forming the desired amide bond. Step 3: By-product Formation The reaction generates hexamethylphosphoramide (HMPA) and benzotriazole (HOBt) as by-products, along with the conjugate acid of the base used. These by-products are generally water-soluble and can be removed during aqueous workup. 2.2 Minimization of Racemization One of the most significant advantages of PyBOP is its ability to minimize racemization of the activated amino acid, particularly at the alpha-carbon of chiral amino acids. The mechanism by which PyBOP achieves low racemization involves several factors: First, the acyloxyphosphonium intermediate is highly reactive and short-lived, reducing the opportunity for side reactions that lead to racemization. Second, the benzotriazoloxy group, once displaced, can act as a weak base but is not sufficiently basic to abstract the alpha-proton under typical coupling conditions. Third, the use of non-nucleophilic bases like DIPEA prevents competing deprotonation pathways that could lead to enolization and subsequent racemization. Comparative studies have shown that PyBOP typically produces less than 0.5% racemization when coupling N-protected amino acids under optimized conditions, which is significantly lower than many other coupling reagents such as DCC or EDC alone. 3. Applications in Synthesis 3.1 Peptide Synthesis PyBOP has become one of the most widely used coupling reagents in both solid-phase peptide synthesis (SPPS) and solution-phase peptide synthesis. Its popularity stems from several key advantages: Solid-Phase Peptide Synthesis (SPPS): In SPPS, PyBOP is particularly valuable for difficult couplings involving sterically hindered amino acids, such as sequential Val-Val, Ile-Ile, or any sequence containing multiple beta-branched residues. The reagent provides consistently high coupling yields (>98%) even for these challenging sequences when used with appropriate activation times (typically 30-60 minutes) and excess reagent (3-5 equivalents relative to resin loading). Solution-Phase Synthesis: For solution-phase peptide synthesis, PyBOP offers the advantage of clean reaction profiles with minimal side products. The water-soluble by-products facilitate purification by simple aqueous extraction, making it ideal for the synthesis of protected peptide fragments that will be further elaborated. Special Applications: PyBOP has proven particularly useful for the synthesis of peptides containing unusual amino acids, including D-amino acids, N-methylated amino acids, and beta-amino acids. The low racemization and high efficiency make it compatible with these sensitive substrates. 3.2 Amide Bond Formation in Drug Discovery Beyond peptide synthesis, PyBOP has found extensive use in medicinal chemistry for the construction of non-peptidic amide bonds. Pharmaceutical companies routinely employ PyBOP in hit-to-lead and lead optimization campaigns where amide formation is required. The reagent’s compatibility with a wide range of functional groups makes it particularly valuable in complex molecule synthesis. Common applications include the coupling of carboxylic acid-containing fragments with amine-containing scaffolds in fragment-based drug discovery, the synthesis of amide-linked heterocyclic compounds, and the preparation of carboxamide derivatives for SAR studies. PyBOP typically provides superior results compared to EDC/HOBt combinations in these contexts, particularly when the substrates contain multiple competing functional groups. 3.3 Macrocyclization Reactions PyBOP has emerged as one of the most reliable reagents for macrolactamization reactions, which are critical for the synthesis of cyclic peptides and macrocyclic drug candidates. The formation of macrolactams often requires highly dilute conditions (typically 0.001-0.01 M) to favor intramolecular cyclization over intermolecular oligomerization. PyBOP excels in these reactions due to its rapid activation kinetics and the low tendency of the activated intermediate to undergo side reactions. Successful macrocyclizations have been reported for ring sizes ranging from 12-membered to >30-membered rings. The reagent is particularly effective when combined with slow addition techniques or pseudo-dilution methods. 3.4 Other Synthetic Applications PyBOP has demonstrated utility in several other synthetic transformations: Esterification: While primarily known for amide formation, PyBOP can also activate carboxylic acids for esterification with alcohols, though this is less common than amidation due to the slower reactivity of alcohols compared to amines. Peptoid Synthesis: PyBOP has been successfully applied to the synthesis of peptoids (N-substituted glycine oligomers), where the absence of an alpha-proton eliminates racemization concerns. Cyclization of Amino Acids: The reagent facilitates the formation of diketopiperazines and other cyclic dipeptide structures through intramolecular amide bond formation. 4. Experimental Considerations and Best Practices 4.1 Optimal Reaction Conditions Achieving optimal results with PyBOP requires careful attention to several experimental parameters: Solvent Selection: PyBOP performs best in polar aprotic solvents. DMF (dimethylformamide) is the most commonly used solvent and generally provides the fastest reaction rates. DCM (dichloromethane) offers the advantage of easier product isolation but may require longer reaction times. Acetonitrile provides a good balance and is preferred when DMF must be avoided. THF can be used but generally gives slower reactions. Base Selection: DIPEA (N,N-diisopropylethylamine, Hünig’s base) is the preferred base for most applications due to its non-nucleophilic character and appropriate pKa. N-methylmorpholine (NMM) is an alternative that sometimes provides faster reactions but may increase the risk of side reactions. The typical base-to-acid ratio is 2-4 equivalents, with excess base ensuring complete deprotonation of the carboxylic acid. Reagent Stoichiometry: For solution-phase reactions, 1.1-1.5 equivalents of PyBOP relative to the carboxylic acid is typically sufficient. For SPPS, 3-5 equivalents based on resin loading are standard. The amine component is often used in slight excess (1.1-1.2 eq) in solution phase or as the limiting reagent in SPPS. Reaction Temperature and Time: Most PyBOP couplings proceed efficiently at room temperature (20-25°C). Reaction times vary from 1-4 hours for solution-phase synthesis to 30-60 minutes for SPPS. Difficult couplings may benefit from extended reaction times or gentle warming to 40°C, though excessive heating should be avoided to prevent racemization and decomposition. 4.2 Handling and Storage PyBOP is hygroscopic and moisture-sensitive, requiring careful handling to maintain its reactivity: The reagent should be stored in a tightly sealed container under an inert atmosphere (nitrogen or argon) at 2-8°C. Desiccants such as Drierite or silica gel should be included in the storage container. Before use, the reagent bottle should be allowed to warm to room temperature before opening to prevent condensation of moisture onto the cold powder. Solutions of PyBOP in DMF or other solvents should be prepared fresh and used within a few hours. Extended storage of solutions leads to gradual decomposition and reduced coupling efficiency. If solution storage is necessary, it should be kept under inert atmosphere at low temperature and used within 24 hours. Visual inspection can help assess PyBOP quality: fresh reagent is white to off-white. Yellowing or browning indicates moisture exposure and oxidation, and such material should not be used for critical coupling reactions. 4.3 Troubleshooting Common Problems Low Coupling Yields: If coupling yields are suboptimal, several factors should be investigated. First, verify the quality of the PyBOP (check for discoloration). Second, ensure that the carboxylic acid starting material is dry, as moisture can lead to hydrolysis of the activated intermediate. Third, confirm that sufficient base is present; inadequate base leads to incomplete activation. Fourth, consider whether the coupling is sterically demanding and whether longer reaction times or additional equivalents of reagent are needed. Racemization Issues: If racemization is observed (detectable by chiral HPLC or NMR analysis of diastereomeric products), the most common causes are excessive base concentration, high reaction temperature, or extended reaction times. Switching to a weaker base, reducing temperature, and minimizing reaction time often resolves the issue. For particularly sensitive substrates, pre-activation protocols where the acid is activated before amine addition may help. Formation of Side Products: The most common side products include N-acyl ureas (from guanidinium impurities in commercial PyBOP), peptide dimers or oligomers (from insufficient dilution during macrocyclization), and dehydration products (from activation of beta- or gamma-amino acids). Purification of commercial PyBOP by recrystallization, use of high dilution techniques, and careful monitoring of reaction progress by LC-MS can mitigate these issues. 5. Comparison with Other Coupling Reagents The peptide coupling reagent landscape is diverse, and selecting the optimal reagent for a given application requires understanding the relative advantages and disadvantages of available options. 5.1 PyBOP vs. HBTU/HATU HBTU (2-(1H-benzotriazol-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate) and HATU (1-[bis(dimethylamino)methylene]-1H-1,2,3-triazolo[4,5-b]pyridinium 3-oxide hexafluorophosphate) are uronium-based coupling reagents that compete directly with PyBOP in many applications. Advantages of PyBOP: PyBOP generally produces lower racemization than HBTU, particularly for couplings involving C-terminal amino acids with acidic side chains (Asp, Glu). PyBOP by-products are easier to remove during workup because they are more water-soluble. PyBOP shows better performance for sterically hindered couplings. Advantages of HBTU/HATU: These reagents typically provide faster reaction kinetics and can complete couplings in shorter times (15-30 minutes vs. 30-60 minutes for PyBOP). HATU in particular shows excellent performance for difficult sequences. They are less expensive on a per-gram basis. Recommendation: For routine peptide synthesis where speed is prioritized and racemization is not a primary concern, HBTU or HATU may be preferred. For high-value sequences, synthesis of peptides with multiple acidic residues, or applications where epimerization must be minimized, PyBOP is the better choice. 5.2 PyBOP vs. EDC/DCC Carbodiimide coupling reagents like EDC (1-ethyl-3-(3-dimethylaminopropyl)carbodiimide) and DCC (N,N’-dicyclohexylcarbodiimide) represent an older class of coupling agents that are still widely used. Advantages of PyBOP: PyBOP provides much higher coupling efficiency without requiring additives (EDC/DCC typically require HOBt or HOAt to achieve acceptable results). PyBOP shows significantly lower racemization. By-product removal is simpler (no insoluble ureas to filter). Advantages of EDC/DCC: These reagents are substantially less expensive. EDC is water-soluble and can be used for aqueous or biphasic coupling reactions. For large-scale industrial synthesis, the cost advantage can be significant. Recommendation: For small-scale, research-grade peptide synthesis or medicinal chemistry applications, PyBOP is superior. For large-scale or process chemistry applications where cost is a major factor, EDC may be preferred despite its drawbacks. 5.3 PyBOP vs. Other Phosphonium Reagents PyBOP belongs to a family of phosphonium-based coupling reagents that includes PyBrOP, PyAOP, and others. PyBrOP (bromotripyrrolidinophosphonium hexafluorophosphate) is structurally very similar to PyBOP but uses bromide instead of benzotriazoloxy as the leaving group. PyBrOP shows similar reactivity and racemization profiles to PyBOP. The main difference is that PyBrOP does not release HOBt as a by-product, which can be advantageous in certain applications where HOBt might cause issues. However, PyBrOP is generally more expensive and less widely available. PyAOP ((7-azabenzotriazol-1-yloxy)tripyrrolidinophosphonium hexafluorophosphate) uses a 7-aza-HOBt-derived leaving group and is claimed to provide even lower racemization than PyBOP in some cases. However, the performance difference is marginal for most applications, and the higher cost limits its use to specialized applications. 6. Practical Protocols 6.1 General Protocol for Solution-Phase Coupling Materials: Carboxylic acid (1.0 equiv), amine (1.1 equiv), PyBOP (1.2 equiv), DIPEA (3.0 equiv), DMF or DCM (solvent), standard glassware, magnetic stirrer. Procedure: To a solution of the carboxylic acid in DMF (0.1-0.2 M) at room temperature under nitrogen, add DIPEA followed by PyBOP. Stir for 5 minutes to pre-activate. Add the amine and stir at room temperature for 1-4 hours. Monitor by TLC or LC-MS. Upon completion, dilute with water or saturated aqueous NH4Cl and extract with EtOAc (3x). Wash combined organic layers with brine, dry over Na2SO4, filter, and concentrate. Purify by flash chromatography or recrystallization. 6.2 General Protocol for SPPS Coupling Materials: Resin-bound amine (1.0 equiv based on loading), Fmoc-protected amino acid (3-5 equiv), PyBOP (3-5 equiv), DIPEA (6-10 equiv), DMF. Procedure: Swell resin in DMF (10 min). Remove Fmoc protecting group with 20% piperidine in DMF (2 × 10 min). Wash resin thoroughly with DMF (5 × 1 min). Prepare coupling solution by dissolving Fmoc-amino acid and PyBOP in minimal DMF, add DIPEA, and immediately add to resin. Agitate for 30-60 min at room temperature. Wash resin with DMF (5 × 1 min). Perform Kaiser test or chloranil test to confirm coupling completion. If incomplete, repeat coupling. Proceed to next cycle or final cleavage. 6.3 Macrolactamization Protocol Materials: Linear peptide with free N-terminus and C-terminal carboxylic acid (1.0 equiv), PyBOP (1.5 equiv), DIPEA (4.0 equiv), DMF. Procedure: Dissolve linear peptide in DMF to achieve a concentration of 0.001-0.005 M (high dilution is critical). Add DIPEA followed by PyBOP. Stir at room temperature for 12-48 hours, monitoring by LC-MS for disappearance of linear peptide and formation of cyclic product. Upon completion, concentrate and purify by preparative HPLC. Alternatively, use pseudo-dilution method: add solutions of peptide, PyBOP, and DIPEA in DMF slowly via syringe pump to a flask containing DMF over 4-12 hours. 7. Current Research Trends and Future Directions 7.1 Solid-Supported and Immobilized Variants Recent research has explored the development of polymer-supported versions of PyBOP-like reagents to facilitate product purification and enable continuous-flow synthesis. These approaches involve covalently attaching phosphonium coupling groups to polystyrene or other polymer supports, allowing the reagent to be removed by simple filtration after coupling is complete. While conceptually attractive, these systems have faced challenges including reduced reactivity compared to solution-phase PyBOP, difficulty achieving complete conversion for sterically hindered substrates, and limited recyclability of the supported reagent. Ongoing research focuses on optimizing linker design and polymer architecture to address these limitations. 7.2 Microwave-Assisted PyBOP Couplings Microwave irradiation has been investigated as a method to accelerate PyBOP-mediated coupling reactions. Several studies have demonstrated that microwave heating can reduce reaction times from hours to minutes while maintaining high yields and low racemization. The technique is particularly valuable for solid-phase peptide synthesis, where microwave-assisted coupling with PyBOP can enable the synthesis of difficult sequences that fail under conventional heating. Temperature control is critical, as excessive heating can lead to increased racemization and side product formation. Typical microwave protocols employ temperatures of 50-75°C for 5-15 minutes. 7.3 Green Chemistry Alternatives The environmental and toxicological concerns associated with HMPA generation and the use of large volumes of organic solvents have motivated research into greener alternatives to PyBOP: Several research groups are developing modified phosphonium reagents that produce less toxic by-products. Others are exploring PyBOP-like reactivity in more sustainable solvents, including ionic liquids, deep eutectic solvents, and aqueous-organic mixtures. While promising, these approaches have not yet achieved the combination of efficiency, low racemization, and broad substrate scope that makes PyBOP so valuable. Enzymatic and biocatalytic alternatives for amide bond formation are also being actively pursued, though these methods currently lack the generality required to replace chemical coupling reagents for complex peptide synthesis. 8. Conclusions PyBOP has established itself as one of the most reliable and versatile coupling reagents in modern synthetic chemistry. Its combination of high reactivity, low racemization, broad substrate scope, and relatively straightforward handling has made it a staple in peptide synthesis laboratories worldwide. While it is not without limitations—including cost, sensitivity to moisture, and generation of potentially problematic by-products—the advantages generally outweigh the drawbacks for most research applications. References 1. Coste, J.; Le-Nguyen, D.; Castro, B. PyBOP: A New Peptide Coupling Reagent Devoid of Toxic By-product. Tetrahedron Lett. 1990, 31, 205-208. 2. Valeur, E.; Bradley, M. Amide bond formation: beyond the myth of coupling reagents. Chem. Soc. Rev. 2009, 38, 606-631. 3. El-Faham, A.; Albericio, F. Peptide Coupling Reagents, More than a Letter Soup. Chem. Rev. 2011, 111, 6557-6602. 4. Montalbetti, C. A. G. N.; Falque, V. Amide bond formation and peptide coupling. Tetrahedron 2005, 61, 10827-10852. 5. Han, S.-Y.; Kim, Y.-A. Recent development of peptide coupling reagents in organic synthesis. Tetrahedron 2004, 60, 2447-2467. 6. Jad, Y. E.; Acosta, G. A.; Naicker, T.; Ramtahal, M.; El-Faham, A.; Govender, T.; Kruger, H. G.; de la Torre, B. G.; Albericio, F. Synthesis and biological evaluation of a teixobactin analogue. Org. Lett. 2015, 17, 6182-6185. 7. Sabatino, G.; Chinol, M.; Paganelli, G.; Pini, A.; Bracci, L.; Papini, A. M. A new biotin derivative-DOTA conjugate as a versatile tool for pretargeted tumor diagnosis and therapy. J. Med. Chem. 2003, 46, 3170-3173. 8. Carpino, L. A. 1-Hydroxy-7-azabenzotriazole. An efficient peptide coupling additive. J. Am. Chem. Soc. 1993, 115, 4397-4398. 9. Albericio, F. Developments in peptide and amide synthesis. Curr. Opin. Chem. Biol. 2004, 8, 211-221. 10. Fmoc Solid Phase Peptide Synthesis: A Practical Approach; Chan, W. C., White, P. D., Eds.; Oxford University Press: Oxford, 2000. |
|
PyBOP
For Research & Development use only. Not for testing and/or use on humans.