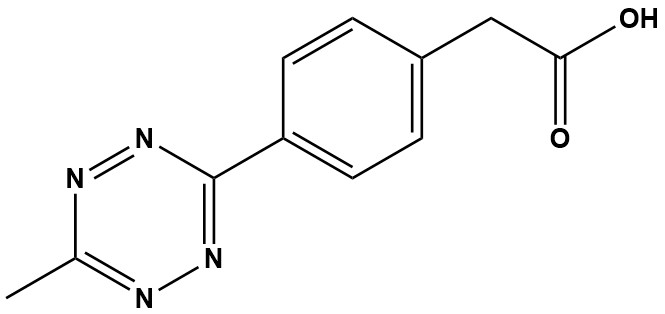
| Synonym: | Methyltetrazine-Phenylacetic Acid |
| CAS #: | 1380500-88-8 |
| Molecular Formula: | C11H10N4O2 |
| Molecular Weight: | 230.2 |
| Methyltetrazine-Acid is a high-performance chemical building block central to the field of bioorthogonal chemistry. It is one of the most popular and stable reagents used for “click chemistry,” specifically for the inverse-electron-demand Diels-Alder (IEDDA) reaction. 1. Chemical Profile and Key Characteristics This molecule is not a pre-activated crosslinker. Instead, it is a versatile building block featuring two key functional groups: • A Methyltetrazine Ring: This is the “click” component. It is a highly strained, electron-poor ring system that reacts with incredible speed and specificity with its reaction partner, trans-cyclooctene (TCO). • A Terminal Carboxylic Acid (-COOH): This is the “handle.” It provides a point of attachment, allowing the tetrazine to be coupled to other molecules (like proteins, antibodies, or peptides) using standard amine chemistry. 2. Mechanism of Action Methyltetrazine-Acid is used in a powerful two-stage bioconjugation strategy. Stage 1: Activation of the Carboxylic Acid The molecule as-sold is not reactive toward proteins. The terminal carboxylic acid (-COOH) must first be activated to make it “amine-reactive.” This is typically achieved using standard carbodiimide chemistry, most commonly with (1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide). • Activation: EDC converts the -COOH group into a highly reactive O-acylisourea intermediate. • Stabilization (Recommended): To improve efficiency and stability in aqueous buffers, Sulfo-NHS (or NHS) is added. Sulfo-NHS intercepts the O-acylisourea to form a much more stable, amine-reactive Sulfo-NHS ester. • Coupling: This activated ester now reacts efficiently with primary amines (−NH2)—such as those on lysine residues of a protein—to form a stable, permanent amide bond. This process allows a researcher to permanently attach the “methyltetrazine” moiety to virtually any protein, antibody, or amine-containing molecule. Stage 2: The Bioorthogonal “Click” Reaction (IEDDA) Once the methyltetrazine is attached to the molecule of interest (e.g., an antibody), it is ready for the click reaction. • Reaction: Inverse-Electron-Demand Diels-Alder (IEDDA) cycloaddition. • Partner: Trans-cyclooctene (TCO) or other strained alkenes. • Speed: This reaction is one of the fastest bioorthogonal reactions known, with second-order rate constants reported as high as 103 to 106 M−1s−1. This speed allows for efficient labeling at very low, physiologically relevant concentrations. • Byproduct: The reaction is exceptionally clean, forming a stable dihydropyridazine adduct and releasing harmless nitrogen gas (N2). • Bioorthogonal: The reaction is “bioorthogonal,” meaning it occurs rapidly in complex biological environments (like cell culture media or in vivo) without cross-reacting with any native biological functional groups (amines, thiols, etc.). 3. Key Advantages and Trade-Offs The primary reason for choosing Methyltetrazine-Acid over other tetrazine derivatives is the critical balance it strikes between reaction speed and stability. 3.1 Advantages • Exceptional Stability: The electron-donating methyl group (-CH3) significantly stabilizes the tetrazine ring. This makes Methyltetrazine-Acid far more resistant to degradation (e.g., hydrolysis or reaction with nucleophiles) than its non-methylated (hydrogen-tetrazine) counterparts. This enhanced stability is crucial for in vivo studies or multi-day experiments. • Extremely Fast Kinetics: While technically slower than the most reactive (and least stable) tetrazines, its reaction with TCO is still incredibly fast, enabling rapid and complete labeling in minutes. • Bioorthogonality: Does not require a toxic copper catalyst (unlike CuAAC “click” chemistry), making it perfect for live-cell imaging and in vivo applications. • Versatility: The -COOH handle allows it to be conjugated to any amine-containing molecule, dye, or surface using well-established EDC/NHS chemistry. • Visual-Readout: The bright red/pink color of the tetrazine disappears upon reaction, providing a simple, visual, and non-destructive way to monitor the reaction’s progress. 3.2 The Stability vs. Reactivity Trade-Off This is the most important consideration when selecting a tetrazine: • Hydrogen-Tetrazine (non-methylated): Maximum Reactivity. This is the fastest-reacting tetrazine. However, it has very poor stability in aqueous solutions and is quickly degraded, making it unsuitable for most biological applications. • Methyltetrazine (this product): High Stability, Very Fast Reactivity. This is the “workhorse” reagent. It gives up a small amount of reaction speed in exchange for the excellent physiological stability needed for reliable bioconjugation. • Di-tert-butyl-tetrazine: Maximum Stability. Extremely stable but also significantly slower to react. For over 90% of applications, including antibody-drug conjugates and live-cell imaging, Methyltetrazine-Acid provides the optimal balance of high stability and extremely fast kinetics. 4. Applications Methyltetrazine-Acid is a foundational tool for advanced bioconjugation: • Antibody-Drug Conjugates (ADCs): An antibody is functionalized with Methyltetrazine-Acid. A separate drug payload is functionalized with TCO. The two are “clicked” together to form the final ADC. • Live-Cell Imaging & Tracking: A TCO-metabolite is fed to cells and incorporated into proteins or glycans. A cell-impermeable Methyltetrazine-fluorophore is then added to the media to “light up” only the cell-surface targets. • PROTACs and Targeted Therapeutics: Used as a stable linker to connect the different components of complex drugs. • Pre-Targeted Imaging (PET/SPECT): A TCO-labeled antibody is injected and allowed to accumulate at a tumor site. Hours or days later, a fast-clearing Methyltetrazine-radioligand is injected, which “clicks” to the antibody at the tumor site for high-contrast imaging. • Surface Functionalization: Used to immobilize proteins or peptides onto TCO-functionalized biosensors or microarray slides. Methyltetrazine-Acid (CAS 1380500-88-8) is a premier bioorthogonal reagent that has revolutionized labeling strategies in chemical biology and biomedicine. Its optimal combination of high IEDDA reactivity, functional handle for conjugation, and favorable stability profile makes it the reagent of choice for demanding applications like pretargeted imaging and live-cell labeling. While considerations regarding its solubility and stability are necessary, its unparalleled speed and specificity solidify its role as an essential tool for researchers developing next-generation diagnostics, therapeutics, and biological probes. |
|
Methyltetrazine-Acid
For Research & Development use only. Not for testing and/or use on humans.