| Synonym: | L-Photo-Leucine |
| CAS #: | 851960-91-3 |
| Molecular Formula: | C5H9N3O2 |
| Molecular Weight: | 143.1 |
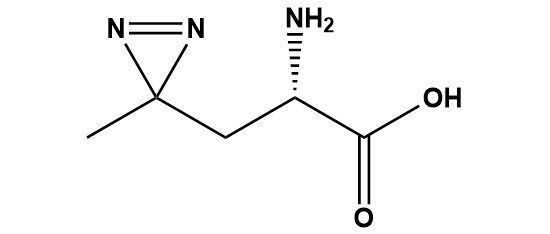
| L-Photo-Leucine (CAS #: 851960-91-3) is a synthetic, photoreactive analog of the essential branched-chain amino acid L-leucine. It belongs to a specialized class of non-canonical amino acids designed for use in photoaffinity labeling (PAL) and photo-crosslinking experiments, which are foundational techniques in chemical biology and proteomics. The compound’s key innovation lies in the replacement of the isobutyl side chain of natural leucine with a diazirine-containing moiety, imparting potent UV-activatable reactivity while preserving the structural similarity to the native amino acid necessary for cellular incorporation. First synthesized in 2005 by Monika Suchanek, Anna Radzikowska, and Christoph Thiele at the Max Planck Institute of Molecular Cell Biology and Genetics in Dresden, L-Photo-Leucine was developed alongside L-Photo-Methionine as part of a pioneering platform for identifying protein-protein interactions (PPIs) using a simple western blot approach. Their landmark publication in Nature Methods transformed how researchers study transient and stable protein complexes in living cells, establishing photo-reactive amino acids as indispensable tools in the chemical biologist’s arsenal. Since its inception, L-Photo-Leucine has found extensive applications across structural biology, drug discovery, target identification, and cell biology, underpinned by its unique capacity to be endogenously incorporated into proteins through normal cellular translation machinery and then photo-activated on demand to trap protein interactions with covalent bonds. 1. Chemical Identity and Physical Properties 1.1 Nomenclature and Identifiers CAS Number: 851960-91-3 IUPAC Name: (S)-2-amino-3-(3-methyl-3H-diazirin-3-yl)propanoic acid Common Name: L-Photo-Leucine; Photo-Leu; H-L-Photo-Leu-OH Molecular Formula: C5H9N3O2 Molecular Weight: 143.1 g/mol MDL Number: MFCD19207670 InChI Key: FWMNROVZZCTELJ-YFKPBYRVSA-N Stereochemistry: S-configuration (L-form only) 1.2 Structural Features L-Photo-Leucine shares the fundamental amino acid scaffold with L-leucine: an alpha-amino group (–NH2), a carboxylic acid group (–COOH), and an alpha-carbon (Cα). The critical structural difference resides in the R-group side chain, where the isobutyl group of native leucine is replaced by a –CH2– methylene bridge leading to a 3-methyl-3H-diazirine ring. The diazirine ring is a three-membered heterocyclic compound consisting of one carbon atom doubly bonded to two nitrogen atoms (C=N–N), which are themselves singly bonded to each other. In L-Photo-Leucine, the diazirine carbon bears a methyl group, making it a 3-methyldiazirine derivative. This compact ring system is critically responsible for all photochemical reactivity of the molecule. The total side chain therefore consists of: Cα–CH2–C(CH3)(=N–N)–, where the terminal C is the diazirine carbon. Notably, only the L-enantiomer (S-configuration) of photo-leucine is synthesized, as all proteinogenic amino acids in humans occur in the L-form. This stereoselectivity is essential for the molecule to bypass the proofreading mechanisms of aminoacyl-tRNA synthetases and be accepted into the ribosomal translation apparatus. 1.3 Chemical Properties Solubility: Soluble in water; recommended to dissolve in water or dilute HCl UV Absorption: Absorption band centered around 345–370 nm (diazirine n→π*) Optimal Photoactivation: ~345–365 nm (UV-A range); 365 nm commonly used experimentally Photostability: Relatively stable under normal white/fluorescent laboratory lighting Storage Conditions: –20°C, protected from light; desiccated conditions recommended 2. Mechanism of Action: Photochemistry of Diazirine 2.1 Ground-State Behavior At ambient temperature and under standard laboratory white-light illumination, L-Photo-Leucine is essentially chemically inert. The diazirine ring, while thermodynamically strained, possesses a kinetic stability that allows the compound to be handled, dissolved, added to cell culture media, and incorporated into proteins without premature activation. This photostability under visible light is one of the key practical advantages of diazirine-based photocrosslinkers compared to earlier aryl azide reagents, which could activate under uncontrolled conditions. 2.2 UV-Induced Photoactivation Upon irradiation with ultraviolet light in the 320–370 nm range (optimally ~345–365 nm), the diazirine ring of L-Photo-Leucine undergoes a concerted photochemical reaction. The electronic excitation of the C=N–N chromophore leads to the homolytic cleavage of both C–N bonds, with the simultaneous loss of molecular nitrogen (N2) as a stable gas. This generates a singlet carbene — an extremely reactive, electron-deficient carbon species with two unpaired electrons — at the former diazirine carbon position within the protein side chain. The singlet carbene is one of the most reactive intermediates known in organic chemistry, with a lifetime estimated in the nanosecond range. In this fleeting window, it reacts indiscriminately with nearby chemical bonds, including: • C–H bonds (via hydrogen abstraction followed by radical recombination or direct insertion) • O–H bonds (insertion) • N–H bonds (insertion) • π bonds such as aromatic rings (cycloaddition) • Heteroatom lone pairs This non-selective reactivity ensures that any molecule within approximately 3–4 Å of the diazirine carbon at the moment of UV irradiation becomes covalently linked. This proximity requirement is central to the high spatial specificity of photo-crosslinking experiments. 2.3 Formation of Stable Covalent Cross-Links When L-Photo-Leucine has been incorporated into protein A, and protein A is engaged in a non-covalent interaction with protein B, the carbene generated from the photo-leucine side chain within protein A can insert into bonds on protein B, creating an irreversible covalent cross-link. The resulting AB complex is stable to denaturing conditions (boiling SDS-PAGE, urea, detergents) and can be subjected to analytical workflows that would otherwise dissociate transient non-covalent interactions. 3. Synthesis and Chemical Preparation 3.1 Original Synthesis (2005, Suchanek et al.) The original synthesis of photo-leucine described in the 2005 Nature Methods paper proceeded through a six-step route starting from commercially available precursors. The synthesis involved: • Preparation of 4,4′-azi-pentanoic acid (the diazirine-containing carboxylic acid precursor) • Reaction with CCl4 and thionyl chloride at 65°C for 30 minutes to install the acid chloride • Alpha-bromination of the azi-carboxylic acid • Aminolysis of the azi-bromo-carboxylic acid to install the amino group • Enzymatic resolution of the racemic intermediate to obtain the L-enantiomer • Final deprotection and salt formation This original route suffered from low overall yield and required enzymatic resolution — a costly, time-consuming step — to achieve stereochemical purity. 3.2 Improved Synthesis (MacKinnon et al., 2007) A significantly improved synthesis was reported by Andrew MacKinnon and colleagues at UCSF in a 2007 JACS paper, reducing the route to fewer steps and improving overall yield substantially. This improved method introduced Boc-(S)-photo-leucine as a key intermediate, prepared via: • Ozonolysis of a commercially available, enantiopure starting material — thereby avoiding enzymatic resolution entirely • Formation of the diazirine using the Church-Weiss methodology (oxidative cyclization of an oxime precursor) • Boc deprotection to yield the free amino acid in high yield and excellent enantiomeric excess The Church-Weiss diazirine-forming reaction involves treating a ketone precursor with hydroxylamine to form the corresponding oxime, followed by amination to give a diaziridine, which is then oxidized (typically with iodine or Ag2O) to give the diazirine ring. This improved route is now the basis for most commercial manufacture of L-Photo-Leucine. 3.3 Protected Derivatives for Peptide Synthesis For incorporation into peptides via solid-phase peptide synthesis (SPPS), N-terminally protected forms are required. Both Boc- and Fmoc-protected L-Photo-Leucine are commercially available. Fmoc-L-Photo-Leucine is particularly widely used for Fmoc-SPPS strategies, which are currently the predominant method in research and industry. The Fmoc group is removed under mildly basic piperidine conditions, which are compatible with the diazirine ring, making Fmoc-Photo-Leucine a tractable building block for automated peptide synthesizers. 4. Biological Incorporation into Proteins 4.1 Mechanism of Cellular Uptake and Incorporation One of the most remarkable properties of L-Photo-Leucine is its ability to be incorporated into proteins by the cell’s own translation machinery. This is possible because the structural similarity of L-Photo-Leucine to L-leucine is sufficient to fool leucyl-tRNA synthetase (LeuRS) — the enzyme responsible for charging leucine onto its cognate tRNA — into accepting photo-leucine as a substrate. Once charged onto tRNA^Leu, the photo-leucine is delivered to the ribosome and inserted into the nascent polypeptide chain wherever a leucine codon (UUA, UUG, CUU, CUC, CUA, CUG) appears. Leucine is one of the most abundant amino acids in human proteins, comprising approximately 9–10% of all residues in the proteome. This high natural abundance means that photo-leucine is incorporated at numerous positions throughout all newly synthesized proteins in treated cells, providing excellent coverage. 4.2 Experimental Protocol for In Vivo Incorporation The standard protocol for incorporating L-Photo-Leucine into cellular proteins involves the following steps: • Culture cells (e.g., HeLa, 293T, COS7, U2OS, A549, A431, HepG2, NIH 3T3, C6) in a leucine-free, methionine-free culture medium (DMEM-LM) supplemented with dialyzed fetal bovine serum • Add L-Photo-Leucine (typically 0.1–1.0 mM) and, optionally, L-Photo-Methionine (another photoreactive analog targeting Met residues) to the leucine-depleted medium • Incubate cells for 16–24 hours (or shorter for pulse labeling) to allow metabolic incorporation • Proceed directly to UV crosslinking without washing if studying in situ interactions It is important to note that photo-leucine incorporation leads to a mixed population of leucine and photo-leucine residues across all newly synthesized proteins. The ratio depends on the concentration of photo-leucine in the medium and any residual leucine present. 4.3 UV Crosslinking of Cellular Samples Following incorporation, cells or cell lysates are irradiated with UV light (345–365 nm) to trigger diazirine activation. Key experimental parameters include: UV Wavelength Recommended Condition: 345–365 nm (365 nm most common) Notes: Long-wave UV; avoids DNA damage from 254 nm germicidal UV Irradiation Time Recommended Condition: 10–15 minutes (in vivo); ~10 min (in vitro) Notes: Longer irradiation may harm cells; rotate samples for uniform exposure Temperature Recommended Condition: On ice (4°C) preferred for in vitro Notes: Reduces thermal decomposition; slows enzymatic reactions during crosslinking Vessel Geometry Recommended Condition: Shallow, uncovered plates Notes: Maximizes UV penetration; covered dishes attenuate UV significantly UV Source Power Recommended Condition: Higher power lamps preferred Notes: Short-wave UV transilluminators or handheld 365 nm lamps commonly used In vivo exposure limit Recommended Condition: ≤15 minutes Notes: Longer UV exposure is cytotoxic; minimize exposure time 5. Research Applications 5.1 Protein-Protein Interaction Studies The primary and most widespread application of L-Photo-Leucine is in the study of protein-protein interactions (PPIs) within living cells. The power of this approach derives from the ability to capture transient interactions — those lasting milliseconds to seconds — that are impossible to detect by traditional co-immunoprecipitation (co-IP) or pull-down assays, which rely on interactions surviving lysis, dilution, and affinity purification steps. In a typical PPI experiment, cells expressing photo-leucine-containing proteins are stimulated with a ligand, hormone, or stress signal (or simply growing under normal conditions), then immediately UV-irradiated to freeze protein complexes in place. After lysis under denaturing conditions, the covalently crosslinked complexes can be immunoprecipitated with antibodies against one partner, separated by SDS-PAGE, and the crosslinked partner identified by western blot, mass spectrometry, or both. 5.2 Photoaffinity Labeling for Drug Target Identification L-Photo-Leucine is extensively used in photoaffinity labeling (PAL) strategies for drug target identification and validation — a critical bottleneck in drug discovery. In this application, photo-leucine is incorporated into a bioactive peptide, cyclodepsipeptide, or peptidomimetic drug molecule (either through SPPS or by metabolic incorporation in bacterial expression systems). When the drug-protein complex is irradiated, a covalent bond forms between the drug and its binding partner, enabling identification of the target even from complex protein mixtures. A landmark demonstration of this approach was the work by MacKinnon et al. (2007), who incorporated photo-leucine into a cyclodepsipeptide inhibitor of cotranslational translocation and used photoaffinity labeling followed by click chemistry with a rhodamine-azide reporter to identify Sec61α — the structural core of the Sec61 translocation channel — as the drug’s molecular target. 5.3 Mapping Protein Interaction Interfaces Beyond simple identification of interacting partners, photo-crosslinking with L-Photo-Leucine can provide structural information about protein interaction interfaces. Because the carbene reacts only with atoms within 3–4 Å of the diazirine carbon at the moment of activation, mass spectrometric analysis of crosslinked peptides can reveal which specific regions of two proteins are in direct spatial proximity. This chemical cross-linking mass spectrometry (XL-MS) approach, using photo-leucine as the crosslinker, provides distance constraints valuable for structural modeling and integrative structural biology. 5.4 Protein-Polymer and Bioconjugation Applications L-Photo-Leucine has been utilized in protein-polymer conjugation strategies. Lin, Boehnke, and Maynard (2014) demonstrated its utility for site-specific protein-polymer conjugation via ligand affinity and photoactivation, specifically conjugating poly(ethylene glycol) (PEG) to glutathione S-transferase (GST) using photo-leucine incorporated into a GST-binding peptide. This approach offers advantages over chemical coupling methods by enabling selectivity driven by molecular recognition before covalent bond formation. 5.5 Receptor Biology and GPCR Studies Photo-leucine-containing peptides have been employed to study G protein-coupled receptor (GPCR) pharmacology. Janz et al. demonstrated direct interaction between an allosteric agonist pepducin and the chemokine receptor CXCR4 using photo-leucine crosslinking, providing direct evidence for a binding mode that was impossible to confirm by biochemical methods alone. This kind of application is particularly powerful for membrane proteins, which are notoriously difficult to study by classical structural approaches. 5.6 Proteome-Wide Interactome Profiling When L-Photo-Leucine is incorporated across the entire proteome of living cells (rather than into a single specific protein), UV irradiation provides a global snapshot of all protein interactions occurring at that moment. Combined with quantitative mass spectrometry (e.g., SILAC or TMT labeling), this proteome-wide photo-crosslinking strategy enables systematic mapping of interactomes under different biological conditions — for example, comparing interactomes of cells before and after drug treatment, differentiation, or infection. 5.7 Study of Protein-Lipid Interactions Diazirines are among the few photoreactive groups capable of reacting with lipid C–H bonds, which are chemically inert to most other reagents. L-Photo-Leucine incorporated into membrane-associated peptides or proteins can therefore be used to probe protein-lipid interactions in native membrane environments, providing information about membrane topology and lipid binding sites. 5.8 Affinity-Based Proteomics (ABP) In tandem with clickable handles (alkyne or azide groups introduced elsewhere in the probe molecule), photo-leucine-containing bifunctional probes support activity-based protein profiling (ABPP) and affinity-based proteomics (ABP). Shi, Zhang, and colleagues demonstrated cell-based proteome profiling of potential dasatinib targets using affinity-based probes incorporating a photo-leucine-like diazirine unit, highlighting the role of these tools in kinome-wide target profiling. 6. Comparison with Related Photoreactive Reagents L-Photo-Leucine Reactive Group: Diazirine (methyldiazirine) Wavelength: 345–365 nm Reactivity: Carbene (non-selective) Key Advantage: Metabolic incorporation; small size Limitation: Random insertion; no sequence selectivity L-Photo-Methionine Reactive Group: Diazirine (methyldiazirine) Wavelength: 345–365 nm Reactivity: Carbene (non-selective) Key Advantage: Met analog; targets Met-rich regions Limitation: Met is less abundant than Leu Benzophenone-Lys Reactive Group: Benzophenone Wavelength: 365 nm Reactivity: Triplet radical (prefers C–H) Key Advantage: Stable; long half-life; can re-shoot Limitation: Bulky; may perturb structure 4-Azido-Phe (AzF) Reactive Group: Aryl azide Wavelength: ~300 nm Reactivity: Nitrene (non-selective) Key Advantage: Genetically encodable (amber suppression) Limitation: UV damages cells; reacts with solvent p-Benzoyl-Phe (pBpa) Reactive Group: Benzophenone Wavelength: 365 nm Reactivity: Triplet diradical Key Advantage: Genetically encodable; efficient Limitation: Bulky phenylalanine analog Diazotrifluoropropionate Reactive Group: Diazoester Wavelength: ~350 nm Reactivity: Carbene Key Advantage: Electrophilic; good for basic residues Limitation: Not amino acid; probe design needed Aryl diazirine (AlkDia) Reactive Group: Aryl diazirine Wavelength: 350–370 nm Reactivity: Carbene Key Advantage: More reactive than alkyldiazirine Limitation: Requires chemical probe design The key competitive advantage of L-Photo-Leucine over benzophenone or aryl azide photoprobes is its metabolic incorporability. Unlike bifunctional chemical crosslinkers that must be added exogenously and may not penetrate cell membranes efficiently, photo-leucine exploits the cell’s own translation machinery to achieve site-specific (all Leu positions) incorporation throughout the proteome. This avoids chemical modification artifacts and allows study of interactions in their native subcellular context. 7. Advantages and Limitations 7.1 Key Advantages • Native cellular context: Incorporation via normal translation means proteins fold correctly and localize normally, unlike chemical crosslinkers added to cell lysates • Stable under white light: Diazirine is photostable under laboratory lighting, allowing easy handling without strict dark-room conditions • Small size: The methyldiazirine group minimally perturbs protein structure compared to bulky benzophenone or biotin tags • Non-selective carbene: The reactive carbene inserts into virtually any chemical bond in proximity, giving high crosslinking efficiency regardless of the functional groups present • High specificity: Only proteins in direct contact (~3–4 Å) at the moment of irradiation are crosslinked — transient interactions are captured with high precision • High protein recovery: ~90% protein recovery reported post-crosslinking in cell lysates • Wide cell line compatibility: Compatible with HeLa, 293T, COS7, U2OS, A549, A431, HepG2, NIH 3T3, C6, and others • Available in protected forms: Boc- and Fmoc-protected derivatives enable incorporation into synthetic peptides via SPPS 7.2 Limitations and Considerations • Indiscriminate reactivity: Because carbenes react with any bond, the crosslinking reaction lacks selectivity for specific amino acid side chains, making it difficult to map the precise crosslink site without high-resolution MS/MS analysis • Competition with endogenous leucine: In cells, photo-leucine competes with endogenous leucine for incorporation; residual leucine must be minimized using leucine-depleted media • UV cytotoxicity: UV irradiation at 365 nm for >15 minutes in living cells can cause DNA damage and cellular stress; experiments must be carefully time-controlled • Incomplete incorporation: Not every leucine residue is replaced by photo-leucine; the stochastic nature of incorporation means some interacting proteins may lack photo-leucine at their interface • Not genetically encodable: Unlike amber-suppression photocrosslinkers (e.g., pBpa, AzF), photo-leucine cannot be site-specifically installed at a defined amino acid position using unnatural amino acid mutagenesis; it incorporates at all Leu positions globally • Potential carbene rearrangement: Singlet carbenes can undergo Wolff rearrangement to ketene, which reacts differently; this side reaction is minimized in protic solvents but should be considered in mechanistic studies 8. Future Perspectives and Emerging Applications The utility of L-Photo-Leucine continues to expand as new analytical methodologies and biological questions emerge. Several frontier areas are likely to drive further use of this compound: 8.1 Integration with Proximity Labeling Emerging approaches combine photo-crosslinking with enzymatic proximity labeling (BioID, TurboID, APEX) for orthogonal validation of PPIs. Photo-leucine crosslinking can confirm direct contacts identified by proximity labeling, adding structural specificity to the spatially resolved but distance-agnostic proximity labeling data. 8.2 In Vivo and Organismal Applications While most photo-leucine experiments have been conducted in cultured cells, there is growing interest in applying metabolic photo-crosslinking in whole organisms — zebrafish, Drosophila, and even rodent models — using systemic delivery of photo-leucine followed by targeted UV irradiation of specific tissues. Such in vivo applications would open new avenues for studying protein interactions in native physiological contexts. 8.3 Cryo-EM and Structural Biology Integration The ability to covalently trap transient protein complexes with photo-leucine crosslinking has significant implications for cryo-electron microscopy (cryo-EM). Crosslinked complexes can be more homogeneous and conformationally stable during grid preparation, potentially enabling cryo-EM structures of otherwise too-dynamic interactions to be determined. Combined with XL-MS data, this could dramatically advance structural understanding of protein interaction networks. 8.4 RNA-Protein and DNA-Protein Interactions The non-selective carbene chemistry of photo-leucine means it can crosslink to RNA and DNA backbones as well as to proteins. This opens the possibility of using photo-leucine-containing RNA-binding proteins to covalently capture RNA-protein and chromatin-associated protein complexes in living cells, extending the application space well beyond classical PPIs. 8.5 Therapeutic Development Although L-Photo-Leucine itself is purely a research tool, insights gained from photo-crosslinking experiments — particularly the structural mapping of PPI interfaces — are invaluable for small-molecule drug design. Fragments, peptides, and macrocycles targeting newly defined interaction surfaces identified through photo-leucine studies represent a pipeline of therapeutic opportunities, particularly for ‘undruggable’ targets where no active site exists. 9. Conclusion L-Photo-Leucine (CAS #: 851960-91-3) stands as a landmark reagent in the field of chemical biology and proteomics. Its elegant design — pairing the cellular recognition of natural L-leucine with the photochemical power of diazirine — has enabled researchers to study protein-protein interactions in their native cellular environment with unprecedented specificity and temporal resolution. From its origins as a tool for simple western blot-based PPI identification to its current roles in structural proteomics, drug target identification, receptor pharmacology, and affinity-based chemical biology, photo-leucine has proven to be a versatile and enduring research tool. Its key strengths — metabolic incorporability, minimal structural perturbation, photostability under ambient light, and non-selective carbene reactivity — collectively make it superior to many alternative photocrosslinkers for in-cell applications. Limitations such as global (non-site-specific) incorporation and competition with endogenous leucine are well-understood and can be managed by careful experimental design. As the fields of structural proteomics, chemical biology, and targeted drug discovery continue to advance, L-Photo-Leucine will undoubtedly remain a cornerstone reagent, enabling new biological discoveries and supporting the development of next-generation therapeutics against some of medicine’s most challenging molecular targets. References 1. Suchanek, M., Radzikowska, A., & Thiele, C. (2005) “Photo-leucine and photo-methionine allow identification of protein-protein interactions in living cells.” Nature Methods, 2(4), 261–267. 2. Hashimoto, M., & Hatanaka, Y. (2008) “Recent progress in diazirine-based photoaffinity labeling.” European Journal of Organic Chemistry, 2008(15), 2513–2523. 3. Kotzyba-Hibert, F., Kapfer, I., & Goeldner, M. (1995) “Recent trends in photoaffinity labeling.” Angewandte Chemie International Edition, 34(12), 1296–1312. 4. Chavez, J.D., et al. (2013) “Chemical crosslinking mass spectrometry analysis of protein conformations and interactions in their native cellular context.” Molecular & Cellular Proteomics, 12(6), 1451–1467. 5. Grunwald, C., et al. (2010) “In vivo quantification of protein-protein interactions inside living cells using fluorescence resonance energy transfer and diazirine-based crosslinking.” 6. Brunner, J. (1993) “New photolabeling and crosslinking methods.” Annual Review of Biochemistry, 62, 483–514. (Historical context on diazirine photochemistry) 7. Iacobucci, C., et al. (2019) “A cross-linking/mass spectrometry workflow based on MS-cleavable, urea-containing crosslinkers and the MeroX software for studying protein structures and protein–protein interactions.” Nature Protocols, 14, 2556–2584. 8. L-Photo-Leucine |
|
H-Photo-Leu-OH
For Research & Development use only. Not for testing and/or use on humans.