| Synonym: | Fmoc-L-Photo-Methionine |
| CAS #: | 945859-89-2 |
| Molecular Formula: | C21H21N3O4 |
| Molecular Weight: | 379.4 |
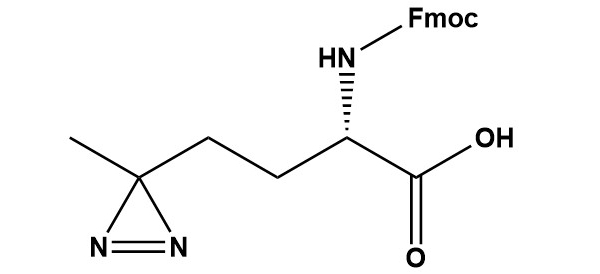
| Fmoc-L-Photo-Methionine (CAS #: 945859-89-2) is an Fmoc-protected, diazirine-bearing photoactivatable analogue of the canonical amino acid L-methionine. The compound incorporates a 3-methyl-3H-diazirine ring at the terminal position of a four-carbon side chain that closely mimics the flexible thioether arm of methionine, generating a highly reactive singlet carbene intermediate upon irradiation with UV-A light (~355–365 nm). This carbene inserts non-selectively into C–H, N–H, O–H, and X–H bonds of proximal molecules, forming covalent adducts that permanently capture transient, non-covalent molecular interactions with zero-length spatial resolution. Co-developed alongside photo-leucine and first described by Suchanek, Radzikowska, and Thiele (Nature Methods, 2005), photo-methionine exploits the relatively permissive substrate specificity of methionyl-tRNA synthetase (MetRS) to enable metabolic incorporation into cellular proteomes, replacing natural methionine residues during protein biosynthesis. The Fmoc-protected derivative critically extends this chemistry to rational, sequence-defined probe design via standard Fmoc solid-phase peptide synthesis (SPPS), enabling placement of the diazirine photophore at any defined position in a synthetic peptide or small-molecule probe. Vila-Perelló, Pratt, Tulin, and Muir (JACS, 2007) further demonstrated that photo-methionine is fully compatible with expressed protein ligation (EPL), enabling site-specific incorporation into large recombinant proteins and semi-synthetic protein constructs — a capability uniquely powerful for interrogating post-translational modification-dependent protein interactions. 1. Introduction and Scientific Context 1.1 The Need for Covalent Capture Tools in Chemical Biology The study of protein–protein interactions (PPIs) and the identification of molecular targets of bioactive small molecules are among the most impactful and technically challenging objectives in modern chemical biology and drug discovery. Traditional biochemical and genetic methods for PPI detection — co-immunoprecipitation, affinity pull-down, yeast two-hybrid, and proximity ligation assays — all share a common vulnerability: they depend on the preservation of non-covalent complexes under conditions that may be far from physiological. Gentle lysis buffers, cold temperatures, and short processing times can only partially mitigate the inevitable perturbation of the molecular interaction landscape that accompanies cell disruption. Photo-crosslinking offers an elegantly simple solution: convert the non-covalent interaction of interest into a stable, covalent bond in situ, before any disruption of the system. A photo-reactive probe is allowed to engage its biological partner through natural non-covalent forces, and then a brief UV pulse triggers the covalent “freeze” of the interaction. The resulting covalent adduct is stable to denaturing conditions, detergent extraction, and boiling SDS-PAGE sample preparation — enabling rigorous biochemical analysis that would be impossible with non-covalent complexes. 1.2 The Diazirine Photophore: Why Methionine? Among the amino acid analogues designed for metabolic photo-crosslinking, the choice of methionine as the structural scaffold for photo-methionine was deliberate and strategically important. Methionine accounts for approximately 2% of average eukaryotic protein sequence composition — less abundant than leucine (~9%), but present in virtually all proteins. Critically, the thioether side chain of methionine (–CH2–CH2–S–CH3) is flexible, moderately hydrophobic, and located at exposed loops, protein–protein contact surfaces, and active site clefts — positions well-suited to forming productive crosslinks with interaction partners. Furthermore, methionine is frequently found at the N-terminus of mature eukaryotic proteins (N-terminal methionine processing), at the initiating Met of each open reading frame, and at conserved functional positions in enzyme active sites (e.g., Met80 in cytochrome c, Met residues in calmodulin binding motifs, and translation initiation Met in peptides). The terminal diazirine ring in photo-methionine is positioned at the location of the methionine sulfur and terminal methyl, maintaining the four-carbon backbone chain length and approximate hydrophobicity of the natural side chain. This geometric and electronic similarity underpins the tolerance of methionyl-tRNA synthetase (MetRS) for photo-methionine and allows its metabolic incorporation in place of methionine in living cells — a critical advantage that distinguishes photo-methionine from most other synthetic photoreactive amino acids. 1.3 Historical Development and the Suchanek 2005 Landmark The concept of using non-canonical amino acids bearing photoreactive groups for in vivo crosslinking of protein complexes was realized practically with the 2005 Nature Methods paper by Suchanek, Radzikowska, and Thiele at the Max Planck Institute of Cell Biology and Genetics in Dresden. Their key insight was that methionine analogs with small aliphatic side chain modifications — specifically, replacement of the thioether by an isosteric diazirine ring — could fool the cellular translation machinery into incorporating the photo-amino acids into proteins. They demonstrated that cells grown in leucine- and methionine-deficient medium supplemented with photo-leucine and photo-methionine incorporated both photo-amino acids efficiently into their proteomes, and that UV irradiation of such cells produced covalent crosslinks between interacting protein pairs that could be detected by SDS-PAGE and western blotting. The technique revealed multiple known protein complexes and — importantly — the previously unknown direct interaction between PGRMC1 and Insig-1, demonstrating the capacity of the technology to generate genuinely new biological knowledge. Subsequently, Vila-Perelló, Pratt, Tulin, and Muir (JACS, 2007) reported an efficient synthesis of photo-Met (the unprotected free amino acid), demonstrated its compatibility with Fmoc-SPPS for site-specific incorporation into synthetic peptides, and used expressed protein ligation (EPL) to produce a semi-synthetic version of the Smad2 signaling protein bearing both photo-Met and a phosphoserine at defined positions. This landmark study demonstrated that photo-Met crosslinking could covalently capture a post-translational modification (PTM)-dependent protein–protein interaction — the MH2–MH2 domain dimerization of phospho-Smad2 — that was otherwise too transient to be captured by conventional means. The Fmoc-protected form of photo-methionine emerged from this synthetic chemistry as the SPPS-compatible building block that enables rational, site-specific photoaffinity probe design. 2. Chemical Identity and Physicochemical Properties 2.1 Nomenclature and Registry Information IUPAC Name: (S)-2-(((9H-fluoren-9-yl)methoxy)carbonylamino)-4-(3-methyl-3H-diazirin-3-yl)butanoic acid CAS Number: 945859-89-2 MDL Number: MFCD31380717 Synonyms: Fmoc-Photo-Met; Fmoc-Photo-Met-OH; Fmoc-L-Photo-Met-OH; (S)-2-(Fmoc-amino)-4-(3H-diazirin-3-yl)pentanoic acid Molecular Formula: C21H21N3O4 Molecular Weight: 379.4 g/mol Stereochemistry: (S)-configuration UV Absorption (λmax): ~265–270 nm (Fmoc); ~345 nm (diazirine n→π*) Photoactivation λ: ~355–365 nm (UV-A) Solubility: DMF, DMSO, DCM (freely); limited aqueous solubility Storage: -20°C, desiccated, protected from light (amber vial or foil-wrapped) 2.2 Structural Architecture: Three Functional Domains The molecule integrates three structurally and functionally distinct components, each essential to its performance as a photo-crosslinking SPPS building block. 2.2.1 The L-Amino Acid Backbone and α-Carboxyl Group The central scaffold of Fmoc-L-Photo-Methionine is a four-carbon α-amino acid with an (S)-configured α-carbon, corresponding to the L-stereoisomer. The free carboxyl group at the α-carbon participates directly in peptide bond formation during SPPS. The absolute (S)-configuration is essential for two reasons: first, it ensures compatibility with standard Fmoc-SPPS protocols and downstream biological applications of the resulting peptide; and second, only the L-form is an efficient substrate for methionyl-tRNA synthetase (MetRS) in metabolic incorporation experiments. The use of the D-form would result in essentially no metabolic incorporation, sharply reducing labeling efficiency in cell-based experiments. 2.2.2 The 3-Methyl-3H-Diazirine Photophore The key distinguishing feature of photo-methionine relative to natural L-methionine is the replacement of the thioether group (–S–CH3) and adjacent methylene (–CH2–) by a 3-methyl-3H-diazirine ring at the terminal end of the four-carbon side chain. The diazirine ring consists of a three-membered C–N=N system in which the central carbon carries a methyl group as its only organic substituent. The ring is reached from the α-carbon via a two-methylene tether (–CH2–CH2–), maintaining a total of four carbons in the side chain — the same chain length as the methionine backbone from Cα to the terminal sulfur. This isosteric relationship is critical: it preserves the approximate molecular volume, flexibility, and hydrophobicity of the methionine side chain, enabling recognition by MetRS and minimizing perturbation of protein folding or binding interface geometry. The diazirine moiety is the smallest known organic photoreactive group. Its molecular mass contribution above a simple methyl group (the two nitrogen atoms add approximately 28 Da) is minimal compared to benzophenone (+182 Da) or aryl diazirine (+144 Da) photophores, explaining why the structural perturbation introduced by photo-methionine incorporation is generally low. The methyl group on the diazirine carbon mimics the terminal methyl of the methionine side chain and serves the additional functions of modulating carbene reactivity and providing steric shielding against intramolecular rearrangements. 2.2.3 The Fmoc N-Protecting Group The 9-fluorenylmethyloxycarbonyl (Fmoc) protecting group is the cornerstone of modern peptide synthesis. Applied to the α-amino group of photo-methionine as a carbamate, it is selectively removed under mild basic conditions (20% piperidine in DMF) through a β-elimination mechanism yielding dibenzofulvene and CO₂. The Fmoc group is orthogonal to the tert-butyl-based (Boc/tBu) acid-labile side-chain protecting groups used in Fmoc-SPPS strategy, enabling selective N-deprotection at each cycle without disturbing the diazirine ring or other protecting groups. After Fmoc removal during SPPS, the diazirine chromophore at ~345 nm persists in the peptide, providing a convenient spectrophotometric handle for confirming incorporation. 2.3 Comparison to L-Methionine L-Methionine differs from photo-methionine exclusively in the terminal two atoms of the side chain: methionine carries –S–CH3 (thioether plus methyl) while photo-methionine carries a –C(CH3)(N=N) diazirine ring at the equivalent position. The key structural analogy is the four-carbon side chain length, the moderate hydrophobicity, and the similar van der Waals profile. The sulfur atom of methionine is replaced by the ring carbon of the diazirine, with the terminal methyl group of methionine preserved as the methyl substituent on the diazirine carbon. This close structural mimicry extends even to the spatial relationships: the distance from the α-carbon to the reactive site (sulfur in Met vs. diazirine carbon in photo-Met) is nearly identical, placing the photophore at precisely the location expected for a methionine-dependent molecular contact. One important distinction from leucine is that the methionine side chain is flexible and linear (no branching at Cγ as in leucine), meaning that the photo-methionine diazirine can adopt a wider range of conformations and reach into different microenvironments than the more constrained photo-leucine. This conformational flexibility can be advantageous for capturing partially occluded binding interfaces, but may also increase the probability of non-specific crosslinking if the side chain samples conformations outside the binding interface. 3. Photochemical Mechanism and Reactivity 3.1 UV Absorption Properties Fmoc-L-Photo-Methionine exhibits two UV absorption regions. The Fmoc chromophore displays a strong π→π* absorption band at approximately 265–270 nm (ε ~ 5,700 M⁻¹cm⁻¹) and a weaker secondary band at 301 nm; both bands disappear upon Fmoc removal during SPPS. The diazirine chromophore absorbs at approximately 340–350 nm (n→π* transition, ε ~ 100–200 M⁻¹cm⁻¹) — a weak but diagnostically useful band that is well-separated from typical protein (280 nm) and nucleic acid (260 nm) chromophores. This diazirine absorption in the UV-A range is preserved after Fmoc removal, persisting in the deprotected peptide and enabling spectrophotometric verification that the photophore was not damaged during synthesis or purification. Irradiation at 355–365 nm selectively excites the diazirine while avoiding excitation of aromatic amino acid residues, minimizing non-specific photochemical damage to the biological system. UV-A sources commonly used for photo-crosslinking include medium-pressure mercury vapor lamps with appropriate bandpass filters, UV-A LED arrays (peak emission 365 nm or 340 nm), and filtered xenon arc lamps. 3.2 The Two-Step Photolysis Mechanism Recent mechanistic work by Zhang et al. (Nature Communications, 2024), combining time-resolved spectroscopy with systematic modulation of irradiation intensity and duration, revealed that alkyl diazirine photolysis — including photo-methionine — proceeds through a sequential two-step mechanism rather than a concerted direct conversion to the free carbene: • Step 1 — Diazo intermediate: Photoexcitation of the diazirine ring causes homolytic ring opening to form a diazo compound (R–C(CH3)=N2). This metastable intermediate absorbs at shorter wavelengths (~340 nm) and is longer-lived than the subsequent carbene. Crucially, the diazo intermediate reacts preferentially with polar, buried residues — particularly acidic residues (Asp, Glu) — through a relatively selective electrophilic mechanism. These buried polar residues are often inaccessible to conventional chemical crosslinkers, making diazo-mediated crosslinking a complementary and unique tool for structural proteomics. • Step 2 — Free carbene: Further photolysis of the diazo compound liberates molecular nitrogen (N₂) and generates a free singlet alkyl carbene. With a lifetime on the nanosecond timescale, this carbene reacts through concerted insertion into virtually any chemical bond in its immediate vicinity (C–H, N–H, O–H, C–C, C–S). The carbene is non-selective and reacts primarily with molecules within direct van der Waals contact (~3–4 Å) at the moment of photolysis. Key Advance: The two-step diazo→carbene mechanism (Zhang et al., Nat. Commun. 2024) means that irradiation parameters can be tuned to favour diazo-selective labelling of buried polar residues (low intensity, longer wavelength) or broad carbene-based crosslinking (high intensity, shorter wavelength). This tunability allows the same photo-methionine probe to generate complementary structural information under different irradiation conditions. 3.3 Carbene Insertion Chemistry The singlet carbene generated from photo-methionine is a highly electrophilic species that reacts through concerted, pericyclic insertion into nearby bonds without generating radical intermediates. Documented insertion reactions include: • C–H bond insertion: The most prevalent reaction in protein environments, forming new C–C bonds with aliphatic, aromatic, and backbone C–H bonds. This non-selectivity is critical for capturing hydrophobic interface contacts. • N–H bond insertion: Reaction with amide backbone N–H bonds, lysine ε-amino groups, arginine guanidinium N–H bonds, histidine imidazole N–H bonds, and tryptophan indole N–H bonds. • O–H bond insertion: Reaction with serine, threonine, and tyrosine hydroxyl groups, and with water molecules (the main competing quenching pathway in aqueous systems). • C–S bond insertion: Relevant for capturing interactions near cysteine thiol groups or other sulfur-containing biomolecules. The inherent proximity of photo-methionine to the sulfur position of natural methionine may enhance C–S insertion reactions with neighboring thioether-containing molecules. • X–H insertion into nucleic acids: Insertion into ribose and deoxyribose C–H bonds and nucleobase N–H bonds, enabling crosslinking to RNA and DNA when the photo-methionine probe is positioned at a protein–nucleic acid interface. 3.4 Reaction Selectivity and Specificity A key consideration for experimental design is the selectivity of photo-methionine crosslinking for genuine binding partners versus non-specific background labeling. Several factors govern this selectivity: • Temporal selectivity: The nanosecond carbene lifetime ensures that only molecules within direct contact distance at the exact moment of photolysis are captured. Dissociated partners, even if transiently dissociated, will not be crosslinked. • Spatial selectivity: Zero-length crosslinks (no linker atoms between the reactive site and the crosslinked residue) provide angstrom-scale spatial information. This is far more restrictive than longer-linker crosslinkers that can capture molecules within 8–15 Å. • Concentration-dependent specificity: Higher-affinity binding partners will spend more time in contact with the photo-methionine probe and will therefore be crosslinked more efficiently than lower-affinity, non-specific interactions. • Diazo intermediate bias: The diazo intermediate preferentially targets acidic and buried polar residues rather than reacting non-specifically with all neighboring molecules, providing an additional layer of selectivity at low irradiation intensities. 3.5 Stability Under Synthesis Conditions The diazirine ring of Fmoc-L-Photo-Methionine is stable under all standard Fmoc-SPPS conditions: piperidine-mediated Fmoc deprotection (20% piperidine/DMF), amino acid coupling with HATU/DIPEA, HBTU/DIPEA, or PyBOP/NMM, side-chain deprotection and resin cleavage with TFA-based cocktails (e.g., TFA/TIS/H2O 95:2.5:2.5, 2–4 h at RT), and reverse-phase HPLC purification in aqueous acetonitrile/TFA or formic acid gradients. This chemical orthogonality allows photo-methionine to be incorporated at any position in a peptide sequence using standard automated or manual synthesis protocols, without modification of the synthesis protocol or special protection schemes. The diazirine ring is similarly stable to the conditions of EPL (thioester ligation, native chemical ligation), enabling semi-synthetic protein construction as demonstrated by Vila-Perelló and Muir. 4. Synthesis and Preparation 4.1 Synthesis of Photo-Methionine: Historical Background The first efficient synthesis of enantiomerically pure photo-methionine was reported by Vila-Perelló, Pratt, Tulin, and Muir (JACS, 2007) as part of their study on Smad2 phospho-dependent oligomerization. Their synthesis started from a suitably protected L-glutamic acid derivative, which provided the (S)-configured α-carbon with high optical purity without requiring enzymatic resolution. The key step involved conversion of a δ-keto intermediate to the diazirine ring via the ammonia / hydroxylamine-O-sulfonic acid (HOSA) oxidative cyclization sequence developed by Church and Weiss. The synthesis was reported in four steps with an overall yield of approximately 32%, representing approximately a sixfold improvement over earlier routes — a decisive improvement that made the compound practically accessible for routine use in chemical biology laboratories. Concurrent work by Durek, Zhang, He, and Kent (Org. Lett., 2007) described the synthesis of photoactive analogues of a cystine knot trypsin inhibitor protein, incorporating diazirine amino acids including photo-methionine via total chemical synthesis, further demonstrating the compatibility of these photo-amino acids with complex peptide synthesis strategies. 4.2 Modern Synthetic Route Contemporary synthesis of Fmoc-L-Photo-Methionine follows the general framework established by Vila-Perelló and Muir, with refinements to improve yield, stereoselectivity, and scalability. The key synthetic steps are: • Preparation of a γ-keto-α-amino acid precursor with the (S)-configuration at Cα, typically from L-glutamic acid or via asymmetric synthesis using chiral auxiliaries. The carboxyl groups are protected as t-butyl esters or benzyl esters throughout the diazirine ring formation steps. • Reaction of the terminal ketone with liquid ammonia under pressure to form an imine (aldimine), which cyclizes in situ with hydroxylamine-O-sulfonic acid (HOSA) to form the diaziridine ring intermediate. • Oxidation of the diaziridine to the diazirine using a mild oxidant (e.g., iodine/triethylamine in DCM, or peroxide-based systems), completing the three-membered C–N=N ring. • Selective removal of all protecting groups from the α-amino and α-carboxyl positions to expose the free amino acid, followed by introduction of the Fmoc group using Fmoc-OSu (Fmoc-N-hydroxysuccinimide ester) in aqueous base/organic co-solvent (e.g., 10% Na₂CO₃ aq./dioxane or acetonitrile). • Purification of Fmoc-L-Photo-Methionine by silica gel column chromatography and/or recrystallization, with quality control by HPLC, NMR, HRMS, and optical rotation. The critical challenge in the synthesis is controlling the oxidation state of the diazirine ring formation step and avoiding over-oxidation or epimerization at the α-carbon. These issues have been largely resolved by using carefully controlled HOSA concentrations, short reaction times, and low temperatures during the oxidation step. Commercial manufacturers have further optimized these protocols for reproducibility and scale. 5. Analytical Characterization 5.1 UV-Visible Spectroscopy UV-Vis spectroscopy provides two key pieces of information for quality control of Fmoc-L-Photo-Methionine. The Fmoc absorption at 265–270 nm confirms presence and relative quantity of the protecting group, while the diazirine absorption at ~340–350 nm confirms integrity of the photophore. A ratio of these two absorptions can be used to assess purity. Following Fmoc removal during SPPS, monitoring the diazirine band at 345 nm provides: (1) confirmation that the photophore survived deprotection; (2) quantification of diazirine-containing peptide in HPLC fractions; and (iii) a handle for optimizing UV crosslinking conditions by measuring the half-life of the chromophore under the specific lamp geometry and irradiation conditions to be used experimentally. 5.2 Nuclear Magnetic Resonance Spectroscopy The 1H NMR spectrum of Fmoc-L-Photo-Methionine in DMSO-d6 shows the following characteristic resonances: • Fmoc aromatic protons: multiplets at δ 7.30–7.90 ppm (8H, aromatic) • NH carbamate proton: broad doublet at δ ~7.60–7.80 ppm (1H, exchangeable with D₂O) • Fmoc 9-methine (CH): triplet at δ ~4.22 ppm (1H) • Fmoc methylene (OCH₂): multiplet at δ ~4.10–4.35 ppm (2H) • α-CH: multiplet at δ ~4.00–4.10 ppm (1H) • β-CH₂: multiplet at δ ~1.80–2.00 ppm (2H, diastereotopic, -CH₂- adjacent to α-carbon) • γ-CH₂: multiplet at δ ~1.70–1.90 ppm (2H, diastereotopic, -CH₂- adjacent to diazirine) • Diazirine methyl (C-CH3): singlet at δ ~1.25–1.35 ppm (3H, characteristic) In 13C NMR, the quaternary diazirine carbon appears as a weak signal at δ ~25–28 ppm, and the diazirine methyl carbon at δ ~19–21 ppm. The α-carbon resonance appears at δ ~52 ppm, and the carbamate and carboxyl carbons at δ ~155 ppm and ~172–174 ppm, respectively. Complete assignment of all carbons, including the fluorene system, is available in the photosupplementary data of the Vila-Perelló et al. (2007) JACS communication. 5.3 Mass Spectrometry ESI-MS (positive mode) of Fmoc-L-Photo-Methionine shows [M+H]⁺ at m/z 380.16, [M+Na]⁺ at m/z 402.14, and [M+K]⁺ at m/z 418.11. High-resolution mass spectrometry (HRMS) confirms the molecular formula C21H21N3O4 with sub-ppm mass accuracy, unambiguously distinguishing photo-methionine from photo-leucine (C20H19N3O4, MW 365.38) by the single additional carbon (one extra methylene in the side chain: four-carbon side chain versus three-carbon in photo-leucine). When incorporated into peptides, LC-MS/MS analysis confirms incorporation through the expected mass addition of 379.14 Da per residue (replacing methionine at 149.05 Da represents a net mass addition of +230.09 Da). In photoaffinity labeling experiments, crosslinked peptide pairs appear as high-mass ions detectable by MALDI-TOF or high-resolution LC-MS, and can be identified using specialized crosslinked peptide search algorithms (e.g., pLink, StavroX, MeroX, or MetaMorpheus with crosslink search capability). 5.4 Chiral HPLC and Optical Rotation Enantiomeric purity of Fmoc-L-Photo-Methionine is confirmed by chiral HPLC (Chiralpak IA or IB column, hexane/ethanol/TFA mobile phase) and optical rotation measurement. The (S)-enantiomer shows a negative optical rotation. Commercial certificates of analysis (CoA) from major suppliers typically include optical rotation data confirming ≥98% e.e. Loss of optical purity (epimerization) at the α-carbon during synthesis is a quality concern that is addressed by chromatographic or recrystallization purification steps. 6. Incorporation Strategies 6.1 Fmoc Solid-Phase Peptide Synthesis (SPPS) The primary designed application of Fmoc-L-Photo-Methionine is as a building block for Fmoc-SPPS. The compound behaves as a drop-in substitute for any Fmoc-amino acid in standard automated or manual synthesis protocols. Dissolved in anhydrous DMF at 0.2–0.5 M concentration (prepared freshly and protected from light), it is activated using standard coupling reagents (HATU, HBTU, or PyBOP) with a base (DIPEA or NMM) and coupled to the growing resin-bound peptide chain. Coupling times of 20–45 minutes are standard; double coupling is recommended for hindered sequences or for critical positions. The ability to position the photo-methionine diazirine at any defined site in a synthetic peptide provides the researcher with the powerful combination of structural control over probe design, precise spatial control over crosslink location, and dramatically simplified mass spectrometric analysis (since only a single crosslinkable site is present). This site-specific approach — enabled by the Fmoc form — is the primary advantage over the global metabolic labeling approach, which distributes photo-methionine across all Met positions in all proteins. Importantly, all SPPS conditions are fully compatible with the diazirine ring. Neither the strongly basic piperidine deprotection step (pH > 10), nor the strongly acidic TFA cleavage step, nor the nucleophilic coupling conditions cause measurable decomposition or photolysis of the diazirine. The only precaution required is protection of all solutions and intermediates from UV light throughout synthesis, handling, and purification. 6.2 Metabolic Incorporation in Living Cells The unprotected free amino acid form, H-L-Photo-Methionine, is incorporated globally into the cellular proteome through metabolic labeling. Methionyl-tRNA synthetase (MetRS) has sufficient substrate promiscuity to charge tRNA^Met with photo-methionine, enabling its ribosomal incorporation in place of methionine during ongoing protein synthesis in living cells. This approach requires no genetic engineering of the host cell and works with essentially all mammalian cell lines tested. The standard dual-labeling protocol involves growing cells in methionine- and leucine-deficient medium supplemented with both photo-methionine (1.7 mM) and photo-leucine (4 mM) simultaneously. The combined use of both photo-amino acids is recommended because their complementary structural coverage (methionine~2% and leucine ~9% of protein composition) together maximize the probability that any given interacting protein pair will have incorporated at least one photoreactive residue at or near the interaction interface. Following metabolic labeling, cells are washed with PBS, placed on ice, and irradiated with UV-A light (310–370 nm) for the appropriate time (determined empirically from the diazirine half-life measurement), then lysed for downstream analysis. 6.3 Expressed Protein Ligation (EPL) for Large Proteins One of the most powerful and distinctive applications of photo-methionine chemistry — enabled specifically by its compatibility with both SPPS and EPL chemistry — is the site-specific incorporation of photo-Met into large, recombinantly produced proteins. In EPL (also called protein semi-synthesis), a synthetic peptide or peptide thioester containing photo-methionine at a defined position (prepared by Fmoc-SPPS) is ligated to a recombinant protein fragment bearing a complementary N-terminal cysteine, forming a native amide bond at the ligation junction. Vila-Perelló et al. (2007) demonstrated this approach masterfully using the Smad2 signaling protein. A synthetic 16-residue C-terminal peptide of Smad2, incorporating photo-Met and a phosphoserine at defined positions, was ligated via EPL to a recombinant truncated Smad2 fragment expressed as a C-terminal intein fusion. The resulting semi-synthetic phospho-Smad2 bearing photo-Met at a specific position was fully functional in biochemical assays and, upon UV irradiation, covalently captured the transient MH2–MH2 domain dimerization interaction in a PTM-dependent manner. This demonstration was the first use of any photo-amino acid to capture a PTM-dependent protein oligomerization event in a site-specific manner — a capability that remains unique to the EPL/SPPS approach using Fmoc-protected photo-amino acid building blocks. 6.4 Bioorthogonal Tag Integration and Bifunctional Probe Design For chemical proteomics applications requiring target enrichment and identification, Fmoc-L-Photo-Methionine is routinely combined with bioorthogonal handles in bifunctional probe designs. A typical probe architecture includes: (1) a target-recognition element (e.g., a bioactive peptide, small molecule pharmacophore, or natural product scaffold); (2) Fmoc-L-Photo-Methionine incorporated at a position expected to contact the binding partner; and (3) a bioorthogonal enrichment tag — either an alkyne (for CuAAC click chemistry with azide-biotin or azide-fluorophore after crosslinking) or a biotin tag (for direct streptavidin enrichment). The alkyne tag approach has the advantage of smaller probe size during the binding/crosslinking step (the click reaction is performed post-crosslinking), while the biotin approach allows immediate enrichment without additional chemistry. Both strategies have been widely validated in the literature. 7. Research Applications 7.1 Discovery of Novel Protein–Protein Interactions in Living Cells The founding application of photo-methionine is the unbiased, proteome-wide discovery of protein–protein interactions in living cells. The landmark Suchanek et al. (2005) study demonstrated that dual metabolic labeling with photo-leucine and photo-methionine, followed by UV crosslinking and biochemical analysis, could capture multiple known and previously unknown protein–protein interaction pairs from COS7 cells. Among the interactions identified was a direct physical interaction between the progesterone receptor membrane component 1 (PGRMC1) and Insig-1 (Insulin-induced gene 1 protein), a component of the sterol regulatory machinery. This interaction was not previously known and had direct implications for understanding cholesterol and lipid metabolism, demonstrating that photo-amino acid crosslinking could generate novel biological knowledge beyond merely confirming known interactions. 7.2 Covalent Capture of PTM-Dependent Protein Interactions One of the most scientifically distinctive applications of photo-methionine — and a key advantage over photo-leucine in this specific context — is the covalent capture of protein interactions that are dependent on post-translational modifications (PTMs). Many signaling-relevant PPIs are conditional on the presence of specific PTMs (phosphorylation, methylation, acetylation, ubiquitination) on one or both interacting partners. These modification-dependent interactions are among the most challenging to study: they are often transient, low-affinity, and present only in specifically activated cellular states. Vila-Perelló, Pratt, Tulin, and Muir (JACS, 2007) addressed this challenge using semi-synthetic Smad2 protein bearing both photo-Met and a phosphoserine at the C-terminal regulatory site. TGF-β signaling activates Smad2 through phosphorylation of two serine residues near its C-terminus (Ser465 and Ser467), inducing dimerization of Smad2 MH2 domains. By incorporating photo-Met at a position within the MH2 domain at the Smad2–Smad2 interface predicted by structural models, and placing the phosphoserine to mimic receptor-activated Smad2, the authors generated a semi-synthetic Smad2 that mimicked the phospho-activated form. UV irradiation of this semi-synthetic protein in the presence of recombinant wild-type Smad2 MH2 domain produced a photo-Met-crosslinked homodimer detected only in the presence of the phosphoserine — directly demonstrating PTM-dependent oligomerization with unprecedented specificity and providing a general template for studying other phosphorylation-dependent protein interactions. 7.3 Drug Target Identification and Chemical Proteomics Photo-methionine-containing photoaffinity probes are increasingly used for drug target identification (target ID) in complex proteomics workflows. A typical approach involves the design of a bifunctional probe in which the pharmacophore of a drug candidate or natural product is coupled (via a linker) to both Fmoc-L-Photo-Methionine and an alkyne for click chemistry enrichment. The probe is incubated with cell lysate or intact cells, allowed to bind its target non-covalently, and then UV-activated to form a covalent adduct. After click chemistry attachment of biotin-azide, streptavidin enrichment, and proteolytic digestion, the crosslinked proteins are identified by quantitative LC-MS/MS. Shi, Zhang, Yao et al. demonstrated this approach for target profiling of the kinase inhibitor dasatinib using a diazirine photoaffinity probe strategy, identifying both known and novel binding proteins in the kinase interactome of living cells. While their specific probe did not use photo-methionine, the same workflow has been successfully adapted for photo-Met-containing peptide probes targeting GPCRs, kinases, and other signaling proteins. A particularly important application is the use of photo-methionine in natural product target deconvolution. Natural products frequently have complex structures that are difficult to modify without losing biological activity; incorporation of a diazirine at a Met-equivalent position in their pharmacophore (or in a peptide that mimics the natural product) provides a minimal-perturbation route to a photoaffinity probe. 7.4 Protein–Polymer and Protein–Material Bioconjugation Lin, Boehnke, and Maynard (Bioconjugate Chemistry, 2014) demonstrated a novel application of photo-amino acid chemistry — specifically GST metabolically labeled with photo-leucine and photo-methionine — for the production of site-specific protein–polymer conjugates. By exploiting the glutathione (GSH) binding specificity of GST to pre-organize the enzyme with a glutathione-functionalized polymer prior to UV crosslinking, the photo-amino acids at the GSH binding interface were selectively crosslinked to the polymer, producing oriented, site-specific protein–polymer conjugates with controlled attachment geometry. This affinity-guided, photo-crosslinking bioconjugation strategy represents an important advance in the production of protein therapeutics, biosensors, and diagnostic reagents, where the orientation and site of polymer attachment profoundly influences protein activity, immunogenicity, and pharmacokinetics. The complementarity of photo-methionine and photo-leucine in this context (together covering Met and Leu residues at the binding interface) enhances crosslinking efficiency compared to using either alone. 7.5 RNA–Protein Interaction Studies Photo-methionine has been exploited for the study of RNA–protein interactions, leveraging the ability of the diazirine carbene to insert into ribose C–H bonds and RNA backbone O–H bonds in addition to protein–protein contacts. Hyun, Han, and Yu (ChemBioChem, 2009) demonstrated that photo-Met-containing amphiphilic α-helical peptides could be photocrosslinked to RNA in a sequence-dependent manner, providing a site-specific approach to studying peptide–RNA binding contacts that is complementary to UV-induced crosslinking using native amino acids (which typically generates crosslinks with uridine and cytidine at shorter UV wavelengths). Additionally, Rentmeister et al. developed photo-Met-based S-adenosylmethionine (AdoMet) analogues for enzymatic transfer of photocrosslinkers to RNA caps, enabling the study of RNA cap-binding protein interactions through diazirine-mediated crosslinking. The diazirine-bearing AdoMet analogue was the best-performing of the three photocrosslinker types tested (diazirine, aryl azide, and benzophenone), as it retained RNA cap-binding affinity (unlike benzophenone) while enabling crosslinking at the less-damaging UV-A wavelength (unlike aryl azide, which requires UV-C). 7.6 Cystine Knot Protein Semi-Synthesis and Structure–Activity Studies Durek, Zhang, He, and Kent (Org. Lett., 2007) incorporated photo-methionine into the cystine knot trypsin inhibitor EETI-II using total chemical synthesis with the Boc-SPPS strategy. By placing photo-Met at specific positions in the reactive loop of this miniprotein, they generated photoactive variants that could covalently capture the trypsin active site upon UV irradiation. This application demonstrated the utility of photo-Met for generating covalent probes of enzyme active sites from constrained peptide scaffolds — a strategy broadly applicable to the growing field of constrained peptide therapeutics, including bicyclic peptides, macrolactams, and stapled helices. 7.7 Characterization of Dimeric Membrane Protein Complexes Hétu, Ouellet, Falgueyret, Ramachandran, Robichaud, and colleagues used photo-leucine and photo-methionine metabolic labeling to demonstrate that cyclooxygenase-2 (COX-2) forms stable dimers and that microsomal prostaglandin E2 synthase-1 (mPGES-1) forms inhibitor-sensitive oligomeric structures in intact cells. This application exemplifies the unique power of photo-amino acid crosslinking for studying membrane protein quaternary structure in a cellular context — a technical challenge for which most conventional biochemical methods are poorly suited due to membrane protein lability after detergent extraction. 7.8 AdoMet Analogue-Based RNA Cap Labeling Photo-methionine-derived S-adenosylmethionine (AdoMet) analogues bearing the diazirine at the methyl-equivalent position (analogues of AdoMet in which the methyl group is replaced by a diazirine) represent a creative extension of photo-Met chemistry to enzymatic photocrosslinking. These AdoMet analogues, first developed by Rentmeister and colleagues, can serve as co-substrates for methyltransferases to site-specifically transfer the diazirine group to RNA cap structures. This chemo-enzymatic approach to RNA photocrosslinking provides unprecedented precision in the placement of the photophore within nucleic acid structures, enabling the study of RNA–protein interactions at single-nucleotide resolution. 8. Comparative Analysis with Related Photo-Amino Acids and Photocrosslinking Agents 8.1 Overview Experimental success in photoaffinity labeling depends critically on choosing the right photocrosslinking agent for the specific application. Photo-Met (alkyl diazirine) Parent amino acid: Methionine Side chain character: Flexible thioether Photophore: Alkyl diazirine Activation λ (nm): 355–365 Reactive intermediate: Singlet carbene Metabolic incorporation: Yes (MetRS) SPPS compatibility: Yes (Fmoc) Structural perturbation: Low-moderate UV cell damage: Low (UV-A) Photo-Leu (alkyl diazirine) Parent amino acid: Leucine Side chain character: Branched aliphatic Photophore: Alkyl diazirine Activation λ (nm): 345–365 Reactive intermediate: Singlet carbene Metabolic incorporation: Yes (LeuRS) SPPS compatibility: Yes (Fmoc) Structural perturbation: Minimal UV cell damage: Low (UV-A) Benzophenone (pBpa) Parent amino acid: Phenylalanine Side chain character: Aromatic Photophore: Benzophenone Activation λ (nm): 350–365 Reactive intermediate: Triplet diradical Metabolic incorporation: Via amber supp. SPPS compatibility: Yes Structural perturbation: High (bulky) UV cell damage: Low (UV-A) Aryl azide Parent amino acid: Phenylalanine Side chain character: Aromatic Photophore: Aryl azide Activation λ (nm): 250–300 Reactive intermediate: Nitrene Metabolic incorporation: Via amber supp. SPPS compatibility: Yes Structural perturbation: Moderate UV cell damage: High (UV-C/B) 8.2 Photo-Methionine versus Photo-Leucine Photo-methionine and photo-leucine are the two founding members of the metabolically incorporable diazirine photo-amino acid family and are most often used in combination. Their complementary properties make them synergistic rather than competitive: • Side chain differences: Photo-Leu has a branched three-carbon side chain (closely mimicking the isobutyl group of leucine), while photo-Met has a flexible four-carbon linear chain (mimicking the thioether arm of methionine). This difference in chain length and flexibility means the two amino acids sample different regions of protein interaction surfaces. • Position coverage: Leucine (~9% of average protein composition) is more abundant and more often found in hydrophobic cores and coiled-coil interfaces, while methionine (~2%) is found at more diverse, often surface-exposed positions including active sites and N-terminal sequences. • MetRS vs. LeuRS tolerance: Photo-Met is a substrate for MetRS; photo-Leu for LeuRS. The two synthetases have different active sites and binding pockets, and the respective tolerances for the diazirine substitution may differ between cell types and conditions. • Structural perturbation: Photo-Leu is generally considered the minimal-perturbation option due to the slightly smaller side chain volume; photo-Met introduces a slightly longer, more flexible photophore that may have marginally greater conformational freedom. • Complementarity in dual labeling: Using both simultaneously, as in the original Suchanek protocol, maximizes proteome coverage and crosslinking probability. For SPPS probe design, the choice between incorporating photo-Met or photo-Leu at a specific position should be guided by the structural context (which natural residue is present in the original sequence and which can be substituted without loss of activity). 8.3 Photo-Methionine versus Benzophenone-Based Crosslinkers Benzophenone photocrosslinking groups — typically incorporated as p-benzoyl-L-phenylalanine (pBpa) via amber suppression — have been widely used in cell biology and structural biology. Relative to photo-methionine, benzophenone crosslinkers offer the advantage of a longer-lived triplet diradical intermediate (microseconds rather than nanoseconds), enabling higher crosslinking yields with longer irradiation times. However, this comes at the cost of significantly greater structural perturbation (the large phenyl-ketone system is much more voluminous than the methyl-diazirine ring), inability to metabolic incorporation in mammalian cells (requiring genetic code expansion), preferential labeling of methionine residues in neighboring proteins through triplet-state C–H abstraction from the methionine thioether — a selectivity that can confound target identification — and significant risk of DNA photodamage due to photosensitization. For experiments requiring minimal structural perturbation, metabolic cell labeling, or study of hydrophobic interfaces lacking nucleophilic residues, photo-methionine is clearly superior. 8.4 Photo-Methionine versus Aryl Azide Photocrosslinking Aryl azide photocrosslinking groups require UV-C or UV-B irradiation (250–310 nm) for activation, which is significantly more damaging to cells, DNA, and proteins than the UV-A irradiation used for diazirine activation. The reactive nitrene intermediates generated by aryl azides are highly reactive but undergo competing ring-expansion reactions that reduce crosslinking selectivity and generate larger structural perturbations at the crosslink site. For these reasons, aryl azides have been largely supplanted by diazirines in chemical proteomics applications where cell viability and biological fidelity are priorities. Photo-methionine is superior to aryl azide crosslinkers for all in-cell applications. 9. Advantages and Limitations 9.1 Key Advantages • Structural mimicry of methionine: The four-carbon diazirine side chain closely reproduces the geometry, flexibility, and hydrophobicity of methionine, enabling metabolic incorporation by MetRS and minimizing perturbation of protein folding and binding interfaces. • Smallest available photophore: The 3-methyl-3H-diazirine is the smallest organic photoreactive group, minimizing the steric perturbation introduced at the modified position compared to benzophenone, aryl diazirine, or aryl azide photophores. • Site-specific incorporation by Fmoc-SPPS: The Fmoc-protected form enables placement of the photophore at any defined position in a synthetic peptide or probe with single-residue precision. • Global metabolic incorporation in living cells: The unprotected free amino acid form is incorporated by native MetRS into cellular proteomes without genetic engineering. • UV-A activation (355–365 nm): Minimal photodamage to cells, DNA, and proteins; compatible with live-cell experiments. • Zero-length crosslinks: Nanosecond carbene lifetime restricts crosslinking to direct molecular contacts, providing angstrom-scale spatial information. • Broad carbene reactivity: Non-selective C–H, N–H, O–H, and C–S insertion captures diverse interface types, including hydrophobic contacts not accessible to amine-reactive crosslinkers. • Compatibility with EPL: Photo-Met is uniquely suited for semi-synthetic protein production via expressed protein ligation, enabling site-specific incorporation into large proteins otherwise inaccessible by total chemical synthesis. • Tuneable mechanism: The two-step diazo→carbene mechanism allows irradiation conditions to be optimized to favor selectivity for buried polar residues (diazo pathway) or broad crosslinking (carbene pathway). • Broad commercial availability: High-purity material available from multiple global suppliers in research quantities. 9.2 Limitations and Challenges • Moderate crosslinking efficiency: Productive crosslinking yields of 10–40% are limited by aqueous carbene quenching, intersystem crossing to triplet carbene, and intramolecular rearrangement pathways. • Selectivity bias toward acidic residues: The diazo intermediate preferentially labels Asp and Glu residues, which can lead to overrepresentation of acidic protein surfaces in crosslinking data and requires careful interpretation of crosslink site assignments. • Lower abundance than leucine: With only ~2% of average protein composition, methionine positions in proteins are fewer and more concentrated than leucine, providing lower statistical probability of having a photo-Met residue at any given binding interface in metabolic labeling experiments. • Limited aqueous solubility of Fmoc form: Organic solvents (DMF, DMSO) are required for the Fmoc-protected building block, which is standard for SPPS but adds operational requirements. • UV equipment requirements: Effective photoactivation at 355–365 nm requires a dedicated UV-A light source (UV-A lamp, LED array, or filtered light source); standard transilluminators designed for ethidium bromide visualization may not provide optimal irradiance. • Strict light exclusion required: Ambient light with UV-A components (fluorescent lamps, sunlight) can cause gradual photolysis of photo-Met-containing compounds during handling, synthesis, and purification. • MS complexity of crosslinked peptides: Identification of crosslinked peptide pairs requires specialized bioinformatics software and careful experimental design to minimize false positive rates. • Longer side chain may increase non-specific crosslinking: Relative to photo-leucine, the longer and more flexible photo-Met side chain may sample more conformations, potentially increasing non-specific background crosslinking if not carefully controlled through low probe concentrations and stringent competition experiments. 10. Storage and Handling 10.1 Storage Conditions Fmoc-L-Photo-Methionine should be stored at −20°C in a sealed, light-protected container (amber-sealed glass vials or foil-wrapped tubes). Moisture should be excluded through desiccant storage or inert gas overlay. Solution stocks in anhydrous DMF (10–100 mM) may be stored at −20°C in amber vials for weeks without significant degradation, but should be prepared freshly where possible. 10.2 Light Management During Experiments All handling of Fmoc-L-Photo-Methionine — including dissolution, SPPS operations, HPLC purification, and incubation with biological samples prior to deliberate UV activation — must be conducted under amber-filtered lighting or in subdued white light with UV-filtering. Commercial photographic amber safelights, amber-tinted fluorescent light covers, or LED-based amber lighting systems are appropriate. UV detection during HPLC should use wavelengths below the diazirine absorption (i.e., 214 nm) to avoid continuous background photolysis of the eluting product. 11. Representative Experimental Protocols 11.1 Incorporation into Synthetic Peptides by Fmoc-SPPS • Prepare a 0.2 M solution of Fmoc-L-Photo-Methionine (CAS 945859-89-2) in anhydrous DMF immediately before use. Store in an amber vial on ice, protected from light. • Set up Fmoc-SPPS resin (e.g., Rink amide MBHA resin for C-terminal amide; Wang resin for C-terminal acid). Perform standard chain assembly using 4 equivalents of each Fmoc-amino acid activated with 3.8 equivalents of HATU and 8 equivalents of DIPEA in DMF (20–45 min coupling per residue). • At the desired position, couple Fmoc-L-Photo-Methionine using the same protocol. Double coupling (consecutive 20 min treatments) is recommended. Perform all steps under amber lighting. • Continue synthesis through Fmoc deprotection cycles (20% piperidine/DMF: 1 × 5 min, 1 × 10 min) and chain elongation. Monitor Fmoc removal by UV absorbance or Kaiser/chloranil test. • After assembly completion, cleave and globally deprotect using TFA/TIS/H₂O (95:2.5:2.5, v/v/v) for 2–4 hours at RT in amber tubes. Precipitate crude peptide with ice-cold diethyl ether (×3 washes). • Purify by RP-HPLC (C18, 5–60% acetonitrile/0.1% TFA, UV detection at 214 nm) under amber lighting. Collect fractions in amber tubes; verify by MALDI-TOF or ESI-MS. • Confirm diazirine integrity by UV absorption at ~345 nm. Calculate concentration from the diazirine ε (~150 M⁻¹cm⁻¹) or amino acid analysis. Lyophilize; store at −20°C protected from light. 11.2 Metabolic Labeling and UV Crosslinking in Mammalian Cells • Grow adherent cells (e.g., COS7, HeLa, HEK293) to 60% confluence in standard medium (DMEM + 10% FBS). • Wash cells ×2 with PBS; replace with Met- and Leu-deficient DMEM + 10% dialyzed FBS. Incubate 30–60 min at 37°C, 5% CO₂ to deplete intracellular amino acid pools. • Add photo-methionine (1.7 mM) and photo-leucine (4 mM) dissolved in DMEM-LM to the deprived cells. Incubate 22–24 hours at 37°C, 5% CO₂. • Remove labeling medium; wash cells ×2 with ice-cold PBS. Cover cells with ice-cold PBS (1–2 mm depth). Transfer plate to ice tray. • Irradiate with UV-A lamp (320–365 nm; e.g., UVP CL-1000, or UV-A LED array at 365 nm, positioned 2–5 cm above cells) for 5–10 minutes on ice. Include three controls: (a) labeled, not irradiated; (b) unlabeled, irradiated; (c) labeled, irradiated (experimental). • Lyse cells immediately in RIPA buffer (1% NP-40, 0.5% sodium deoxycholate, 0.1% SDS, 50 mM Tris pH 7.4, 150 mM NaCl, protease inhibitors). Clarify by centrifugation (14,000 × g, 10 min, 4°C). • Analyze by SDS-PAGE, western blotting, co-immunoprecipitation, or submit to LC-MS/MS-based proteomics workflow. 11.3 Photo-Methionine-Based EPL for Site-Specific Labeling of Large Proteins • Synthesize a C-terminal peptide thioester containing photo-Met at the desired position using Fmoc-SPPS on a thioester-generating resin (e.g., safety-catch resin) or using the N-acyl benzimidazolinone (Nbz) thioester precursor strategy. • Express the complementary protein fragment (containing a C-terminal intein fusion for thioester generation or N-terminal Cys for direct NCL) in E. coli using standard recombinant expression. • Perform native chemical ligation (NCL) between the protein thioester and the synthetic Cys-peptide containing photo-Met in NCL buffer (6 M Gdn·HCl, 200 mM sodium phosphate pH 7.0, 50 mM MPAA or TFET as thiol additive, Ar atmosphere). React 24–48 h at RT. • Purify the semi-synthetic protein by Ni-NTA affinity chromatography (if His-tag is included), size-exclusion chromatography, or RP-HPLC. Verify by intact protein mass spectrometry. • Use for UV photocrosslinking experiments in vitro (in binding assays, with purified interaction partners) or in semi-cellular reconstitution systems. 12. Recent Advances and Emerging Applications 12.1 Mechanistic Understanding of the Two-Step Pathway The most significant recent advance in understanding photo-methionine chemistry — and alkyl diazirine chemistry generally — is the mechanistic elucidation of the two-step diazo→carbene photolysis pathway, published in Nature Communications (2024) by Zhang and colleagues. Using an in-line system with systematic modulation of UV light intensity and irradiation time, they demonstrated that the diazo intermediate preferentially labels buried polar residues (especially Asp, Glu) that are inaccessible to conventional chemical crosslinkers, while the carbene provides the broad, non-selective crosslinking for which diazirines are known. This mechanistic insight opens the possibility of using irradiation parameter control as an experimental lever to obtain complementary structural information from a single photo-Met probe — a technique not previously available. 12.2 Integration with Quantitative Chemical Proteomics Photo-methionine photocrosslinking is increasingly integrated with advanced quantitative proteomics platforms, including isobaric labeling (TMT-10plex, TMTpro-16plex) and stable isotope labeling by amino acids in cell culture (SILAC), enabling dose-response competitive profiling of binding proteins across entire cellular proteomes. These workflows identify primary targets, off-targets, and concentration-dependent interactions with statistical rigor, providing multi-dimensional target engagement landscapes for drug candidates that are far more informative than simple binary target identification. 12.3 Photo-Met-Inspired AdoMet Analogues for RNA Biology The structural similarity of photo-methionine to methionine has inspired the development of photo-Met-based S-adenosylmethionine (AdoMet) analogues as cosubstrates for RNA methyltransferases. These analogues allow chemo-enzymatic installation of a diazirine group at specific positions within RNA molecules — particularly at mRNA cap structures (N7-methylguanosine). Upon UV irradiation, the installed diazirine crosslinks to the cap-binding protein, enabling the study of RNA–protein interactions with nucleotide-level spatial resolution. This emerging application combines enzymatic site-specificity with photo-crosslinking covalent capture in a way that has no precedent in conventional RNA biology. 12.4 Semi-Synthetic Protein Drug Design Using Photo-Met The EPL-compatible photo-Met chemistry pioneered by Vila-Perelló and Muir is now being applied in the context of biologics discovery. Semi-synthetic proteins carrying photo-Met at defined positions within therapeutic domains (e.g., cytokine receptor-binding loops, antibody CDRs, enzyme active site peptides) can serve as photocrosslinking affinity reagents for target engagement studies, antibody epitope mapping, and receptor structure characterization. As constrained peptide therapeutics (stapled peptides, bicyclics, macrocycles) advance toward clinical translation, photo-Met incorporation into these scaffolds provides a valuable tool for confirming target engagement in complex biological matrices. 12.5 Cryo-EM Validation Using Photo-Met Crosslinks The zero-length crosslinks generated by photo-methionine are being applied as orthogonal distance restraints for cryo-EM structure validation. Because photo-Met crosslinks report direct contact distances (< 4–5 Å between the diazirine carbon and the crosslinked residue), they provide much tighter structural constraints than conventional bis-NHS crosslinkers (11–15 Å range). Crosslink data from photo-Met experiments are being incorporated into integrative structural modeling workflows — combining cryo-EM density maps with MS-derived crosslink restraints and computational docking — to improve the precision of protein complex structural models, particularly for flexible or heterogeneous assemblies where cryo-EM density maps alone are insufficient. 12.6 Spatial Proteomics: Compartment-Specific Crosslinking Photo-methionine is being integrated with organelle-specific proximity labeling (APEX2, BioID) to enable spatial proteomics workflows that characterize protein interaction networks within defined subcellular compartments. By combining a compartment-targeting enzyme (which labels the local protein neighborhood by proximity) with metabolic photo-Met incorporation (which captures direct contacts), researchers can obtain layered information: first, the identity of proteins in a given cellular locale, and second, which of those proteins are in direct physical contact. This hierarchical approach generates uniquely informative datasets for understanding organelle biology and membrane protein organization. 13. Conclusions and Outlook Fmoc-L-Photo-Methionine (CAS 945859-89-2) is an indispensable tool in the chemical biology toolkit — a compound whose deceptively simple structural modification of the methionine side chain (replacement of –S–CH3 by a 3-methyl-3H-diazirine ring) unlocks an extraordinary range of experimental capabilities spanning from whole-cell interactomics to single-site semi-synthetic protein chemistry. Its ability to be metabolically incorporated into living cell proteomes by native MetRS without genetic engineering is a defining advantage that most other photocrosslinking agents cannot match. The Fmoc-protected form extends this versatility to rational probe design through SPPS, enabling hypothesis-driven experiments with single-residue precision. The compatibility of photo-Met with expressed protein ligation — uniquely powerful for capturing PTM-dependent protein interactions in large proteins — remains a hallmark capability that continues to find new applications as the field of post-translational modification biology expands. Looking forward, several converging developments will further elevate the impact of Fmoc-L-Photo-Methionine chemistry. The mechanistic understanding of the diazo→carbene two-step pathway opens new possibilities for selectivity-controlled crosslinking through irradiation parameter tuning. Integration with quantitative chemical proteomics (isobaric labeling, SILAC) and spatial proteomics (APEX2, BioID) is generating increasingly nuanced target engagement landscapes. The development of photo-Met-based AdoMet analogues is extending diazirine chemistry into RNA biology in wholly new directions. And the application of photo-Met crosslinks as cryo-EM validation restraints places this chemistry at the frontier of integrative structural biology. As chemical biology, structural proteomics, and drug discovery continue to converge in their need for precise, minimally perturbative tools for studying molecular interactions in native environments, Fmoc-L-Photo-Methionine stands as a compound whose full potential remains to be realized — one whose foundational chemistry, established in 2005, continues to generate new scientific insights nearly two decades later. References 1. Suchanek M, Radzikowska A, Thiele C. Photo-leucine and photo-methionine allow identification of protein-protein interactions in living cells. Nat Methods. 2005;2(4):261–267. DOI: 10.1038/nmeth752. [Original description of photo-Met technology and metabolic incorporation] 2. Vila-Perelló M, Pratt MR, Tulin F, Muir TW. Covalent capture of phospho-dependent protein oligomerization by site-specific incorporation of a diazirine photo-cross-linker. J Am Chem Soc. 2007;129(27):8068–8069. DOI: 10.1021/ja072013j. [Efficient synthesis of photo-Met; SPPS compatibility; EPL semi-synthesis; Smad2 phospho-dependent crosslinking] 3. Durek T, Zhang J, He C, Kent SBH. Synthesis of photoactive analogues of a cystine knot trypsin inhibitor protein. Org Lett. 2007;9(26):5497–5500. DOI: 10.1021/ol702461z. [Total chemical synthesis of photo-Met containing miniprotein probes] 4. Hyun S, Han A, Yu J. Photocrosslinking of RNA and PhotoMet-containing amphiphilic alpha-helical peptides. ChemBioChem. 2009;10(6):987–989. DOI: 10.1002/cbic.200900100. [RNA–protein crosslinking with photo-Met peptides] 5. Lin EW, Boehnke N, Maynard HD. Protein-polymer conjugation via ligand affinity and photoactivation of glutathione S-transferase. Bioconjug Chem. 2014;25(10):1902–1909. DOI: 10.1021/bc500380r. [Oriented protein-polymer bioconjugation using photo-amino acids] 6. Yang T, Liu Z, Li XD. Developing diazirine-based chemical probes to identify histone modification readers and erasers. Chem Sci. 2015;6(2):1011–1017. DOI: 10.1039/C4SC02328E. [Epigenetics application of diazirine probes] 7. Janz JM, Ren Y, Looby R, et al. Direct interaction between an allosteric agonist pepducin and the chemokine receptor CXCR4. J Am Chem Soc. 2011;133(39):15878–15881. DOI: 10.1021/ja206661w. [GPCR photoaffinity labeling; SPPS photo-Leu/Met probe] 8. Kleiner RE, Hang LE, Molloy KR, Chait BT, Kapoor TM. A chemical proteomics approach to reveal direct protein-protein interactions in living cells. Cell Chem Biol. 2018;25(1):110–120.e3. DOI: 10.1016/j.chembiol.2017.10.001. [Chemical proteomics with diazirine probes] 9. Das J. Aliphatic diazirines as photoaffinity probes for proteins: recent developments. Chem Rev. 2011;111(8):4405–4417. DOI: 10.1021/cr1002722. [Comprehensive review of aliphatic diazirine chemistry] 10. Hill JR, Robertson AAB. Fishing for drug targets: a focus on diazirine photoaffinity probe synthesis. J Med Chem. 2018;61(16):6945–6963. DOI: 10.1021/acs.jmedchem.7b01561. [Synthetic strategies for diazirine probes] 11. Halloran MW, Lumb JP. Recent applications of diazirines in chemical proteomics. Chem Eur J. 2019;25(20):4885–4898. DOI: 10.1002/chem.201805004. [Chemical proteomics applications review] 12. Zhang Z, et al. Dissecting diazirine photo-reaction mechanism for protein residue-specific cross-linking and distance mapping. Nat Commun. 2024;15:6083. DOI: 10.1038/s41467-024-50315-y. [Two-step diazo→carbene mechanism elucidation] 13. Kannt A, Bhatt DL, Landmesser U, Lüscher TF. Expanding druggable space through photoaffinity labeling. Nat Rev Drug Discov. 2024. [Emerging directions in PAL drug discovery] 14. West AV, Woo CM. Design and evaluation of a cyclobutane diazirine alkyne tag for photoaffinity labeling in cells. J Am Chem Soc. 2022;144(46):21174–21183. DOI: 10.1021/jacs.2c08257. [Next-generation diazirine PALBOX photophore] |
|
Fmoc-Photo-Met-OH
For Research & Development use only. Not for testing and/or use on humans.