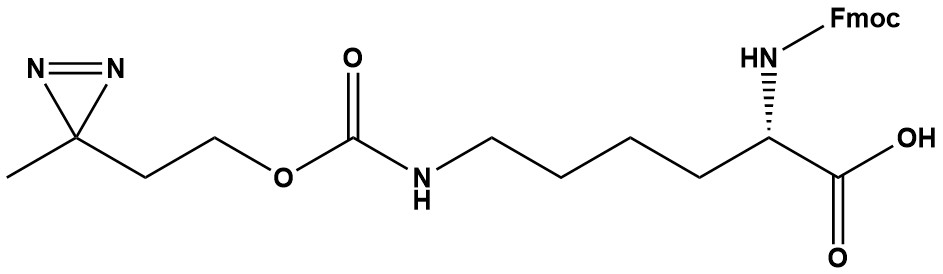
| Synonym: | Fmoc-L-Photo-Lysine |
| CAS #: | 2250437-42-2 |
| Molecular Formula: | C26H30N4O6 |
| Molecular Weight: | 494.5 |
| Fmoc-L-Photo-Lysine (CAS #: 2250437-42-2) is a synthetic, photoreactive unnatural amino acid derivative widely employed in chemical biology, proteomics, and structural biochemistry. Its defining feature is the diazirine functional group incorporated onto the lysine side chain, enabling covalent crosslinking with proximal biomolecules upon ultraviolet irradiation. 1. Chemical Identity and Nomenclature CAS Number: 2250437-42-2 Molecular Formula: C26H30N4O6 Molecular Weight: 494.5 g/mol IUPAC Name: (S)-2-(((9H-fluoren-9-yl)methoxy)carbonylamino)-6-((2-(3-methyl-3H-diazirin-3-yl)ethoxy)carbonylamino)hexanoic acid MDL Number: MFCD31380716 Synonyms: Fmoc-Photo-Lys, Fmoc-L-Photo-Lys-OH, Fmoc-Photo-Lys-OH 2. Structural Features The molecule integrates three functionally distinct components into a single building block: The Fmoc (9-fluorenylmethyloxycarbonyl) group protects the alpha-amino group at the N-terminus of the lysine backbone. The Fmoc group is a protecting group commonly used in solid-phase peptide synthesis. It allows for the selective incorporation of Fmoc-L-Photo-Lysine into peptides at specific positions while protecting other functional groups on the amino acid side chain. The diazirine moiety is appended to the epsilon-amino group of lysine via a propoxy-carbamate linker. Upon UV irradiation (approximately 360 nm) for 10 minutes on ice, the diazirine yields a highly reactive carbene species that rapidly reacts with neighboring molecules to form an irreversible covalent bond. The L-lysine scaffold provides the stereochemically defined (S-configuration) backbone that can be treated like a canonical amino acid during peptide assembly. 3. Mechanism of Action The photochemical mechanism proceeds in two key stages: Stage 1 — UV Activation: When exposed to UV light, the diazirine group undergoes a rearrangement to produce a carbene. A diazirine moiety was chosen over other photoaffinity labels due to its reported short activation time, high stability, and low background photo cross-linking. Stage 2 — Covalent Crosslinking: The generated carbene is highly electrophilic and reacts non-selectively but in a proximity-dependent manner. This carbene can react with nucleophiles such as amines, thiols, and other functional groups present in proteins or peptides, leading to covalent crosslinking. Recent mechanistic studies have revealed additional nuance. Alkyl diazirine photolysis proceeds through a two-step pathway involving sequential generation of diazo and carbene intermediates. The diazo intermediate preferentially targets buried polar residues, many of which are inaccessible with known chemical crosslinking probes. Furthermore, alkyl diazirines exhibit preferential labeling of acidic amino acids in a pH-dependent manner that is characteristic of a reactive alkyl diazo intermediate, and probes with a net positive charge tend to produce higher labeling yield. 4. Synthesis The synthesis of Fmoc-L-Photo-Lysine typically involves several steps: first, protection of the lysine alpha-amino group using the Fmoc group, followed by introduction of the diazirine moiety through a reaction involving diazomethane or similar reagents. Construction of Fmoc-protected photo-reactive amino acid building blocks generally requires multi-step synthesis with an overall synthetic yield of approximately 20%. More streamlined approaches using phenyliodonium diacetate (PIDA)-mediated transformation have been reported as alternatives that reduce step count and improve yields for related diazirine-containing building blocks. 5. Applications in Research 5.1 Photoaffinity Labeling (PAL) and Chemical Proteomics Fmoc-L-Photo-Lysine is a diazirine-containing, Fmoc-protected lysine amino acid and multifunctional photo-crosslinker. Its incorporation into peptides or small-molecule probes and tools allows for photoaffinity labeling of cellular targets and protein-protein interactions upon UV light (~360 nm) irradiation to form a covalent bond. Photo-affinity labeling has become one of the most powerful strategies to study protein-protein interactions (PPIs). Upon photoirradiation, photocrosslinkers generate highly reactive species that react with adjacent molecules, resulting in a direct covalent modification. 5.2 Protein–Protein Interaction Studies By incorporating Fmoc-L-Photo-Lysine into peptides or small-molecule probes, researchers can use UV light to trigger the formation of covalent bonds with nearby proteins, allowing scientists to identify protein interaction partners and study protein-protein interaction dynamics. The spatial and temporal precision afforded by light activation is a core advantage of this approach. 5.3 Drug Target Identification and Validation Diazirine-based photoaffinity probes can facilitate the drug discovery process by covalently capturing transient molecular interactions. This can help identify target proteins and map the ligand’s interactome. Multifunctional probe building blocks such as Fmoc-L-Photo-Lysine continue to accelerate drug discovery research for probing cellular mechanisms, target identification and validation, and understanding traditionally undruggable targets. 5.4 Natural Product Target Profiling A generally applicable protocol for the photoactivated immobilization of unmodified and microgram quantities of natural products on diazirine-decorated beads enables systematic affinity-based proteome profiling. Among diverse tested molecules, 25 of 31 could be successfully immobilized using this approach. 5.5 Receptor Capture and GPCR Biology All receptors with an extracellular domain can be captured in intact cells using a photo-reactive group. A washing step after ligand–receptor binding but before receptor capture can be introduced, thus reducing nonspecific binding and background labeling by the probe. 5.6 Histone PTM Reader Profiling Diazirine-based histone peptide probes can recruit specific readers of histone post-translational modifications (hPTMs), with the diazirine-based photoreaction converting transient protein–protein interactions into irreversible covalent bonds to capture those readers. 6. Advantages Over Competing Photocrosslinkers Traditional photocrosslinkers used in PAL include benzophenone, aryl azide, and diazirine. Upon photoirradiation, these photocrosslinkers generate highly reactive species that react with adjacent molecules, resulting in a direct covalent modification. Among these, diazirines offer several advantages: • Compact size: Diazirines are among the smallest photoreactive groups that form a reactive carbene upon light irradiation. Their small footprint minimizes perturbation of the biomolecule’s native binding behavior. • High photostability: Diazirine groups are highly stable in the dark and in ambient conditions, supporting robust probe shelf life and experimental workflows. • Low background: Compared to other photoaffinity labels, the diazirine moiety has a reported short activation time, high stability, and low background photo cross-linking. • Biocompatibility: Fmoc-L-Photo-Lysine is well-tolerated by cells, making it suitable for studying protein interactions in living systems. • Orthogonality: The Fmoc protecting group allows selective deprotection during SPPS without disturbing the diazirine functionality. 7. Use in Solid-Phase Peptide Synthesis (SPPS) Fmoc-L-Photo-Lysine is fully compatible with standard Fmoc-based SPPS workflows. It is incorporated as a building block at defined positions during stepwise peptide assembly, using standard coupling reagents (e.g., HATU, PyBOP) in DMF or NMP. The diazirine moiety is stable to the piperidine-mediated Fmoc deprotection conditions (~20% piperidine in DMF) and to the acidic TFA-based cleavage conditions typically used to release peptides from Wang or Rink amide resins. Standard handling precautions — working under subdued or amber light and avoiding unnecessary UV exposure — are sufficient to preserve the diazirine intact throughout synthesis. 8. Comparison with the Unprotected Analog An unprotected version of photo-lysine (without the Fmoc group) is used for direct incorporation into proteins via genetic code expansion or metabolic labeling strategies in cultured cells. Fmoc-L-Photo-Lysine (CAS 2250437-42-2), by contrast, is the building block of choice for chemical synthesis — enabling precise, site-specific placement of the photocrosslinker at any position within a synthetic peptide or small-molecule probe, without reliance on cellular machinery. 9. Summary Fmoc-L-Photo-Lysine (CAS #: 2250437-42-2) is a powerful, synthetically versatile building block that bridges the disciplines of peptide chemistry and chemical biology. Its dual functionality — Fmoc protection for SPPS compatibility and a diazirine group for UV-triggered covalent crosslinking — makes it uniquely suited for constructing photoaffinity probes with precise spatial control. Its small size, photostability, and biocompatibility position it as a preferred reagent for mapping protein–protein interactions, identifying drug targets in complex proteomes, and probing the cellular interactome of both synthetic peptides and small-molecule probes. As chemical proteomics and target-ID workflows continue to mature, Fmoc-L-Photo-Lysine remains a cornerstone reagent in the toolbox of modern chemical biologists and medicinal chemists. References 1. Yang et al., Nat. Chem. Biol. 2015 — photo-lysine for capturing proteins that bind lysine post-translational modifications. 2. Suchanek, Radzikowska & Thiele, Nature Methods 2005 — photo-leucine and photo-methionine for identifying protein–protein interactions in living cells. 3. Janz et al., J. Am. Chem. Soc. 2011 — direct interaction between an allosteric agonist pepducin and chemokine receptor CXCR4. 4. Lin, Boehnke & Maynard, Bioconjugate Chem. 2014 — protein–polymer conjugation via ligand affinity and photoactivation of glutathione S-transferase. 5. West et al., J. Am. Chem. Soc. 2021 — labeling preferences of diazirines with protein biomolecules. 6. Jiang et al., Nat. Commun. 2024 — dissecting diazirine photo-reaction mechanism for residue-specific crosslinking and distance mapping. |
|
Fmoc-Photo-Lys-OH
For Research & Development use only. Not for testing and/or use on humans.