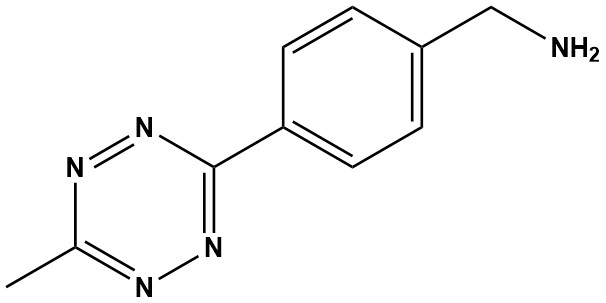
| CAS #: | 1345955-28-3 |
| Molecular Formula: | C10H11N5 |
| Molecular Weight: | 201.2 |
| Methyltetrazine-Amine (CAS 1345955-28-3) is a specialized, high-performance chemical building block used in bioorthogonal chemistry. Unlike the NHS-ester (which reacts with amines) or the Acid (which must be activated to react with amines), this molecule features a primary amine (-NH2). This functionality makes it a nucleophilic reagent. It is designed to react with electrophilic groups (such as activated esters, isothiocyanates, or epoxides) to “install” the methyltetrazine click handle onto a target molecule, surface, or scaffold. 1. Mechanism of Action Methyltetrazine-Amine operates through a two-stage process: Conjugation followed by Click Reaction. 1.1 Conjugation (The Installation Step) The primary amine group is used to attach the tetrazine moiety to a molecule of interest. Because it is a nucleophile, it reacts with: • NHS Esters: To form stable amide bonds. This is the most common application (e.g., reacting with an NHS-functionalized fluorescent dye or PEG chain). • Activated Carboxylic Acids: Using chemistry to couple the amine to a protein or peptide carboxyl group. • Epoxides and Isothiocyanates: To form secondary amines or thioureas, respectively. 1.2 The “Click” Reaction (The Application Step) Once the molecule is labeled with the tetrazine, it undergoes the Inverse-Electron-Demand Diels-Alder (IEDDA) reaction. • Partner: Trans-Cyclooctene (TCO). • Process: The Tetrazine attacks the strained TCO ring. • Result: A dihydropyridazine linkage is formed, and nitrogen gas (N2) is released. • Speed: This is one of the fastest bioconjugation reactions known (k2 up to 30,000 M−1s−1), making it viable for low-concentration biological tracking. 2. Key Advantages 2.1 Stability vs. Reactivity Balance The “Methyl” in Methyltetrazine is functional, not just structural. • Hydrogen-Tetrazines (H-Tet): Extremely fast but chemically unstable in water/serum. • Methyl-Tetrazines (Me-Tet): The methyl group donates electron density, slightly lowering the reaction speed compared to H-Tet, but drastically increasing stability in physiological buffers and serum. 2.2 Versatility in Linker Synthesis This amine is the “universal adapter” for creating new tetrazine reagents. Researchers use it to synthesize: • Tetrazine-Maleimide: By reacting Methyltetrazine-Amine with a bifunctional SMCC crosslinker. • Tetrazine-DBCO: For dual-click applications. • Tetrazine-Drug Conjugates: Attaching the click handle to drug payloads that have carboxylic acid handles. 2.3 Bioorthogonality The tetrazine group is inert toward naturally occurring biological functional groups (amines, thiols, hydroxyls). It reacts only with strained alkenes/alkynes (like TCO), allowing for high-specificity labeling in complex environments like cell lysates or whole organisms. 3. Primary Applications • Antibody-Drug Conjugates (ADCs): Used to synthesize the linker payload. The amine group attaches to the drug (or a spacer), leaving the tetrazine exposed to “click” onto a TCO-labeled antibody. • Surface Functionalization: Used to modify carboxylated surfaces (e.g., biosensor chips, beads, or nanoparticles). Once the surface is coated with tetrazines, it can efficiently capture TCO-tagged biomolecules from solution. • Live Cell Imaging: A TCO-modified sugar or amino acid is metabolically incorporated into a cell. A dye labeled with Methyltetrazine-Amine is then added. The dye “clicks” to the cell surface or intracellular target instantly, with low background. • Radiolabeling: Used to attach chelators (like DOTA or NOTA) to tetrazines for Pre-targeted PET imaging. The chelator is attached via the amine, and the construct is clicked to an antibody in vivo. 4. Handling and Storage • Storage: Store at -20°C. It is crucial to keep the product desiccated and protected from light. Tetrazines can degrade upon prolonged exposure to UV/ambient light. • Solution Stability: Stock solutions should be prepared in anhydrous DMSO or DMF. These stocks are generally stable for weeks at -20°C if kept dry. Avoid storing in aqueous buffers for long periods, as the amine can oxidize or the tetrazine can slowly degrade over time. Methyltetrazine-Amine (CAS 1345955-28-3) is an indispensable and powerful tool in the modern chemical toolbox. Its unique bifunctional design, combining a highly reactive and bioorthogonal tetrazine core with a versatile amine linker, makes it a cornerstone for constructing sophisticated molecular architectures. Its primary role in enabling ultra-fast, catalyst-free IEDDA ligations has revolutionized strategies in bioconjugation, pretargeted radiochemistry, and live-cell imaging. |
|
Methyltetrazine-Amine
For Research & Development use only. Not for testing and/or use on humans.