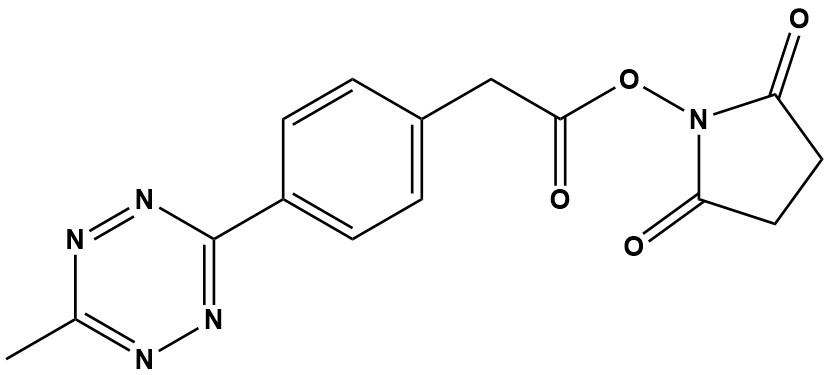
| CAS #: | 1644644-96-1 |
| Molecular Formula: | C15H13N5O4 |
| Molecular Weight: | 327.3 |
| Methyltetrazine-NHS ester (CAS Number 1644644-96-1) is one of the most popular and highly efficient amine-reactive bioorthogonal reagents used in chemical biology. It is a derivative of Methyltetrazine-Acid, with the crucial difference being that its carboxylic acid group has been pre-activated into a highly reactive -hydroxysuccinimide (NHS) ester. This pre-activation makes the reagent immediately ready for one-step conjugation with primary amine groups (-NH2) found on lysine residues and the -termini of proteins. After conjugation, the molecule retains the stable and highly reactive Methyltetrazine moiety, which serves as a powerful “click” handle for subsequent reactions. 1. Chemical Profile and Key Characteristics • Chemical Name: 2,5-Dioxopyrrolidin-1-yl 2-(4-(6-methyl-1,2,4,5-tetrazin-3-yl)phenyl)acetate • Function: One-step conjugation of the Methyltetrazine “click” handle onto primary amines. • Core Reaction: Inverse-Electron-Demand Diels-Alder (IEDDA) cycloaddition. • Solubility: Highly soluble in polar aprotic solvents such as Dimethyl Sulfoxide (DMSO) and Dimethylformamide (DMF). It has very limited stability and solubility in aqueous buffers and should only be dissolved immediately before use. 2. Mechanism and Application Methyltetrazine-NHS ester is designed for sequential, two-step bioconjugation: Step 1: Amine Conjugation (The “Labeling” Step) The NHS ester is highly electrophilic and reacts rapidly and specifically with primary amines (-NH2) present on the target molecule (e.g., an antibody, protein, or peptide) in aqueous buffers at pH 7.0–9.0. • Result: Formation of a stable, covalent amide bond. • Benefit: This pre-activated form eliminates the need for separate EDC or HATU activation steps required when using the non-activated Methyltetrazine-Acid. This simplifies the protocol, reduces potential side reactions, and is often preferred for large, sensitive biomolecules. • Consideration: The NHS ester is sensitive to hydrolysis (reaction with water), especially at higher pH. The reagent must be dissolved immediately before use. Step 2: The Bioorthogonal “Click” Reaction (The “Clicking” Step) Once the protein or molecule is successfully labeled with the Methyltetrazine moiety, it can be “clicked” with its partner, trans-cyclooctene (TCO). • Reaction: The inverse-electron-demand Diels-Alder (IEDDA) reaction. • Kinetics: This is one of the fastest bioorthogonal reactions known, often reaching rate constants up to 106 M−1s−1. • Specificity (Bioorthogonality): The reaction is highly selective, occurring rapidly and efficiently in complex biological media (e.g., cytoplasm, serum) without interfering with native biomolecules like amines or thiols. • Clean Byproduct: The reaction forms a stable dihydropyridazine adduct and releases harmless nitrogen gas (N2). 3. Advantages over Other Tetrazine Reagents Methyltetrazine-NHS ester is the preferred choice for many researchers due to its balance of stability, reactivity, and ease of use. 3.1 Superior Stability (vs. Hydrogen-Tetrazines) The methyl group (-CH3) attached to the tetrazine ring provides a stabilizing effect, protecting the ring from degradation, hydrolysis, and reaction with nucleophiles in biological media. This is essential for applications requiring long incubation times or in vivo use. 3.2 One-Step Activation (vs. Methyltetrazine-Acid) • Methyltetrazine-Acid requires a carbodiimide (like EDC) and an activator (like Sulfo-NHS) to form an amine-reactive intermediate. This is a two-step activation process that can be complex and introduce impurities. • Methyltetrazine-NHS ester is pre-activated. This makes it a ready-to-use, single-reagent system for protein labeling, ensuring higher consistency and purity in the final conjugate. 3.3 Versatility (vs. Sulfo-NHS versions) While often used in its water-soluble ester form, this non-sulfonated NHS ester is soluble in organic solvents (like DMSO or DMF) and typically membrane-permeable (or at least less restricted than its charged sulfo-counterpart). This allows for labeling intracellular proteins or molecules that cannot be easily accessed by charged reagents. 4. Primary Applications Methyltetrazine-NHS ester is widely used across advanced biological and chemical disciplines: • Antibody-Drug Conjugates (ADCs): The primary use is to functionalize antibodies through their lysine residues, providing a stable handle for subsequent “clicking” of a TCO-functionalized cytotoxic drug payload. • Protein Labeling and Tracking: Attaching the Methyltetrazine handle to proteins or peptides, which can then be clicked to a TCO-functionalized fluorescent dye for imaging (e.g., in flow cytometry, Western blotting, or microscopy). • Cell Surface Modification: Labeling proteins on the exterior of living cells for downstream applications like cell-cell tracking or probing receptor interactions. • Hydrogel & Material Functionalization: Using the tetrazine handle to efficiently immobilize proteins or biomolecules onto TCO-modified surfaces or polymeric materials. • Sequential Click Chemistry: Used in combination with other click systems (like CuAAC or SPAAC) for multi-functional labeling due to its high specificity. 5. Handling and Storage • Storage: Should be stored desiccated at −20°C, protected from moisture and light. • Preparation: The reagent is typically dissolved in anhydrous organic solvent (like DMSO or DMF) immediately before use to form a concentrated stock solution. • Use: The stock solution is then diluted into a cold, non-amine containing aqueous buffer (e.g., PBS or HEPES) for the conjugation reaction. The final working concentration should be used immediately to minimize hydrolysis of the reactive NHS ester. Methyltetrazine-NHS ester is a ready-to-use amine-reactive reagent for installing a methyltetrazine bioorthogonal handle on proteins, peptides, surfaces, or small molecules. It couples via standard NHS chemistry (avoid amine buffers), then enables rapid, catalyst-free IEDDA ligation with TCO/BCN probes for imaging, pretargeting, ADC/linker assembly and materials functionalization. While its cost and sensitivity to hydrolysis are important considerations, the benefits of a streamlined, high-yielding and reproducible protocol make it an indispensable asset in the modern chemical biology toolkit. |
|