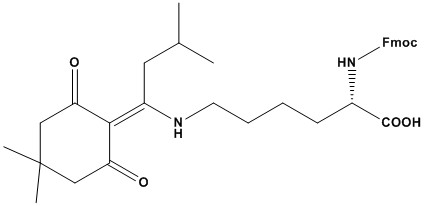
| Synonym: | Fmoc-Nε-1-(4,4-dimethyl-2,6-dioxocyclohex-1-ylidene)-3-methylbutyl-L-lysine |
| CAS #: | 204777-78-6 |
| Molecular Formula: | C34H42N2O6 |
| Molecular Weight: | 574.7 |
| Fmoc-Lys(ivDde)-OH is a protected derivative of the amino acid lysine, specifically designed for use in solid-phase peptide synthesis (SPPS), particularly in Fmoc (fluorenylmethyloxycarbonyl) chemistry. It is widely used for incorporating lysine into peptide sequences while allowing selective deprotection of the lysine side chain. Below is a detailed review of this compound, including its properties, applications, and considerations: Fmoc-Lys(ivDde)-OH is a building block for peptide synthesis that incorporates lysine with orthogonal protection for its ε-amino side chain. The ivDde (1-(4,4-dimethyl-2,6-dioxocyclohexylidene)-3-methylbutyl) group is selectively removable under mild conditions, enabling further modifications or conjugations during synthesis. 1. Chemical Structure (1). Fmoc Group: The Fmoc (fluorenylmethyloxycarbonyl) group protects the α-amino group of lysine during synthesis. It is removed under basic conditions (e.g., piperidine). (2). ivDde Group: The ivDde (1-(4,4-dimethyl-2,6-dioxocyclohexylidene)-3-methylbutyl) group protects the ε-amino side chain of lysine. It is selectively removed using hydrazine or hydroxylamine, leaving other protecting groups intact. (3). Lysine Backbone: The compound includes the lysine amino acid core, with a carboxylic acid group for coupling to the growing peptide chain. 2. Properties (1). Solubility: Fmoc-Lys(ivDde)-OH is soluble in polar organic solvents such as DMF (dimethylformamide) and DMSO (dimethyl sulfoxide), making it suitable for SPPS. (2). Stability: The Fmoc group is stable under acidic conditions but can be removed under basic conditions. The ivDde group is stable under both acidic and basic conditions but is selectively cleaved using hydrazine or hydroxylamine. (3). Molecular Weight: The molecular weight of Fmoc-Lys(ivDde)-OH is approximately 535.63 g/mol. (4). Purity: High-purity Fmoc-Lys(ivDde)-OH is commercially available, ensuring reliable and reproducible results in peptide synthesis. 3. Applications (1). Peptide Synthesis: Fmoc-Lys(ivDde)-OH is used to incorporate lysine into peptide sequences during Fmoc-based SPPS. Lysine is a common amino acid in peptides due to its positive charge and role in protein-protein interactions. (2). Selective Deprotection: The ivDde group allows for selective deprotection of the lysine side chain, enabling further modifications such as conjugation with labels, tags, or other functional groups. (3). Branched Peptides: Fmoc-Lys(ivDde)-OH is often used in the synthesis of branched or multivalent peptides, where the ε-amino group of lysine serves as a branching point. (4). Therapeutic Peptides: Lysine-containing peptides are used in drug development, particularly for antimicrobial peptides and peptide-based vaccines. Fmoc-Lys(ivDde)-OH ensures the correct incorporation of lysine without side reactions. (5). Bioconjugation: The selectively deprotected lysine side chain can be used for site-specific bioconjugation, such as attaching fluorescent dyes, biotin, or PEG chains. 4. Advantages (1). Selective Deprotection: The ivDde group can be selectively removed under mild conditions (e.g., hydrazine), enabling further modifications without affecting other protecting groups. (2). Orthogonal Protection: The ivDde group is orthogonal to other protecting groups, such as Fmoc and Boc, allowing for versatile synthetic strategies. (3). High Purity: Commercially available Fmoc-Lys(ivDde)-OH is typically of high purity, ensuring reliable and reproducible results. (4). Versatility: The selectively deprotected lysine side chain allows for a wide range of applications, including bioconjugation and branched peptide synthesis. 5. Limitations (1). Deprotection Conditions: The ivDde group requires hydrazine or hydroxylamine for removal, which may not be compatible with all peptide sequences or functional groups. (2). Steric Hindrance: The bulky ivDde group can sometimes cause steric hindrance during coupling reactions, particularly in sequences with multiple lysine residues. 6. Comparison with Other Lysine Derivatives (1). Fmoc-Lys(Boc)-OH: Uses the Boc (tert-butoxycarbonyl) group for side chain protection, which requires stronger acidic conditions (e.g., TFA) for removal. (2). Fmoc-Lys(Mtt)-OH: Uses the Mtt (4-methyltrityl) group, which can be selectively removed under mild acidic conditions. (3). Fmoc-Lys(Alloc)-OH: Uses the Alloc (allyloxycarbonyl) group, which can be removed under neutral conditions using palladium catalysts. Among these, Fmoc-Lys(ivDde)-OH is particularly valued for its selective deprotection under mild conditions and orthogonality to other protecting groups. Fmoc-Lys(ivDde)-OH is a critical reagent for incorporating lysine into peptides during Fmoc- based SPPS. Its ability to selectively deprotect the lysine side chain under mild conditions makes it a preferred choice for applications requiring further modifications, such as bioconjugation and branched peptide synthesis. However, its steric hindrance, and requirement for specific deprotection conditions should be considered when planning experiments. Overall, Fmoc- Lys(ivDde)-OH is an indispensable tool for synthesizing lysine-containing peptides in both research and therapeutic applications. References: 1. Chhabra, S. R., et al. (1998). “A Novel Orthogonal Protection Scheme for Solid-Phase Peptide Synthesis Using the ivDde Group.” Tetrahedron Letters, 39(10), 1603-1606. 2. Chandra, K., et al. (2014). “A Tandem In Situ Peptide Cyclization through Trifluoroacetic Acid Cleavage.” Angewandte Chemie International Edition, 53(36), 9450-9455. 3. Peptide Synthesis 4. Overview of Solid Phase Synthesis 5. Fmoc Solid Phase Peptide Synthesis 6. Boc Solid Phase Peptide Synthesis 7. Solid-phase peptide synthesis: from standard procedures to the synthesis of difficult sequences |
|
Fmoc-Lys(ivDde)-OH
For Research & Development use only. Not for testing and/or use on humans.